Home
> Musings: Main
> Archive
> Archive for May - August 2022 (this page)
| Introduction
| e-mail announcements
| Contact
Musings: May - August 2022 (archive)
Musings is an informal newsletter mainly highlighting recent science. It is intended as both fun and instructive. Items are posted a few times each week. See the Introduction, listed below, for more information.
If you got here from a search engine... Do a simple text search of this page to find your topic. Searches for a single word (or root) are most likely to work.
Introduction (separate page).
This page:
2022 (May - August)
August 31
August 24
August 17
August 10
August 3
July 27
July 20
July 13
July 6
June 29
June 22
June 15
June 8
June 1
May 25
May 18
May 11
May 5
Also see the complete listing of Musings pages, immediately below.
All pages:
Most recent posts
2026
2025
2024
2023:
January-April
May-December
2022:
January-April
May-August: this page, see detail above
September-December
2021:
January-April
May-August
September-December
2020:
January-April
May-August
September-December
2019:
January-April
May-August
September-December
2018:
January-April
May-August
September-December
2017:
January-April
May-August
September-December
2016:
January-April
May-August
September-December
2015:
January-April
May-August
September-December
2014:
January-April
May-August
September-December
2013:
January-April
May-August
September-December
2012:
January-April
May-August
September-December
2011:
January-April
May-August
September-December
2010:
January-June
July-December
2009
2008
Links to external sites will open in a new window.
Archive items may be edited, to condense them a bit or to update links. Some links may require a subscription for full access, but I try to provide at least one useful open source for most items.
Please let me know of any broken links you find -- on my Musings pages or any of my web pages. Personal reports are often the first way I find out about such a problem.
August 31, 2022
Briefly noted... Effect of rocket launches on the atmosphere
August 31, 2022
Combustion leads to pollution. The details depend on the nature of the fuel and the conditions; the overall effect also depends on the amount. A recent article looks at the effect of rocket launches on the atmosphere, using theoretical calculations. The big message is that an increasing frequency of rocket launches could well lead to them becoming a significant source of both carbon dioxide and nitrogen oxides.
* News stories:
- Rocket engine exhaust pollution extends high into Earth's atmosphere -- Understanding rocket emissions in the atmosphere by modeling fluid dynamics of rocket exhaust gases. (Science Daily (American Institute of Physics, the journal publisher), May 17, 2022.)
- Atmospheric Pollution from Rockets. (University of Nicosia, undated.) Includes links to some of the other news coverage.
* The article: Atmospheric pollution from rockets. (Ioannis W Kokkinakis & Dimitris Drikakis, Physics of Fluids 34:056107, May 2022.) Check Google Scholar for a freely available copy. At this writing, one of the available copies is the final published version.
More about elephants, cancer, and p53
August 30, 2022
In an earlier post, we noted that elephants have 20 copies of the p53 gene [link at the end]. Since p53 is a tumor suppressor, that could help explain why elephants have a low incidence of cancer. A recent article goes further, suggesting that elephants may have better p53 genes than we do.
The scientists have gene sequences and expected protein structures for the various p53 isoforms in elephant. They differ, which per se should be no big surprise; multiple copies of a gene may diverge. What's interesting is the properties of the various forms of elephant p53.
The following figure gives an example...
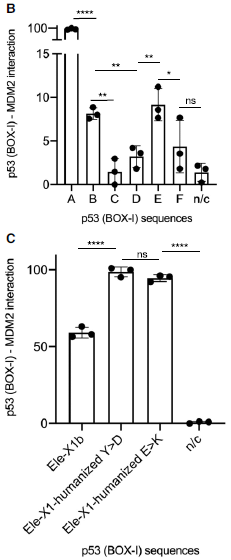
|
The elephant p53 isoforms are grouped into six types, called A though F. (The bar "n/c" is for a negative control.) Examples of those p53 forms were tested for how well they bound to a protein called MDM2 (We'll discuss the importance of this particular binding later.)
In part B (top), the amount of binding is shown on the y-axis; note the split scale. The form that bound the best (A) was set to 100%; the binding for the others is relative. They vary, but forms B-F gave less than 10% binding, compared to A.
Part C (bottom) adds another tidbit. The A form of elephant p53 is very similar to human p53. They differ at two amino acids in the region of the protein being studied here. The scientists changed them, one at a time, to the human amino acid. The graph in part C shows that both of these amino acid changes, making the elephant protein more "humanized", led to better binding to MDM2. (Again, the binding values are relative; the highest value is set to 100%.)
The testing is done with a key peptide from p53, not the whole protein.
This is part of Figure 3 from the article.
|
The general point is that the collection of p53 isoforms in elephant vary in how well they bind to MDM2.
So what? Well, MDM2 plays a key role in the stability of p53. When MDM2 binds to p53, it initiates a process that leads to the degradation of p53. (The p53-MDM2 interaction has multiple roles.)
So it would seem that elephants not only contain more p53 than we do, but that much of their p53 is more stable than ours. Further, the range of properties of the elephant p53 isoforms makes one wonder what other differences there are in function. We might wonder what we will learn about human p53 -- and human cancer -- by further study of the diversity of the elephant p53 family.
A caution... p53 is complicated. Even in humans, with only one gene for p53, there are multiple forms of the protein, due to processes such as alternative splicing. Learning more about p53 is good, but one shouldn't expect practical benefits to be easy.
The tests shown above were done with elephant p53 proteins and human MDM2 proteins. What if elephant MDM2 was used? Wouldn't that be more logical? They had reasons for doing it their way (MDM2 is very similar in both), but did limited testing using the homologous system. It does matter. That leads to some reservation about the details shown above. Nevertheless, the work suggests that the elephant p53 isoforms are different, and probably more stable. That is the important point for now. We also note that the article contains extensive computer modeling that supports the general conclusion; in fact, the computer work is the heart of the article.
News stories:
* Elephant genes could hold the key to avoiding cancers, study -- New insights into molecular interactions which could help people become less prone to cancer. (Pranjal Mehar, Tech Explorist, July 15, 2022.)
* Elephant genes could hold the key to avoiding cancers. (University of Oxford, July 15, 2022.)
The article, which is open access: The Elephant Evolved p53 Isoforms that Escape MDM2-Mediated Repression and Cancer. (Monikaben Padariya et al, Molecular Biology and Evolution 39:msac149, July 2022.)
Background post: Why do elephants have a low incidence of cancer? (March 20, 2016). The article of this earlier post is the first item in the reference list of the current article.
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Cancer. It includes an extensive list of relevant Musings posts.
If your computer was powered by photosynthesis, would you have to water it?
August 29, 2022
Let's set the question in the title aside for the moment, and look at the premise: a computer powered by photosynthesis. That development is reported in a recent article.
In a sense, it is not a big deal. Computers run on electricity. Various sources of electricity can be used. We plug the computer in, or use batteries. We know that living cells generate electricity. Bacterial cells. Microbial fuel cells are a thing. We feed them organic fuel (such as sugar), and they provide electricity, which can be used. What's new here is to use photosynthetic bacteria. Put the device by the window, the bacteria use the light and make electricity, which can power the computer.
* The "computer" in the current work is just a single microprocessor, a type of device increasingly found in the Internet of Things.
* Why not just use a solar cell? Hold that question for now. But one good answer is that the goal is simply to try new things.
Here's the device to generate solar bio-electricity for a computer. This is the compact version.

|
That's it. It's a small culture chamber, with access for light.
The most important feature not visible here is that the chamber also contains an aluminum mesh, which serves as an electrode.
The authors describe the device as being about the size of a AA battery. An AA battery is 50 mm long and 14 mm in diameter. So the new device is a bit bigger, especially for cross-sectional area, but the battery gives you a reasonable sense.
This is Figure 7 from the Electronic Supplementary Material accompanying the article.
|
There are various lab tests reported, but then something closer to a real-world test...
The test is done in a private residence. The device is positioned in front of a window; looks like it is sitting on an inverted drinking glass.
The device is connected to some electronics, which have been programmed to consume power. For example, the microprocessor may compute for 45 minutes, then idle for 15 minutes, with the cycle repeated continuously during the 24 hour day.
This is Figure 10 from the Electronic Supplementary Material.
|

|
Here are some data from that test...

The three graphs each show one parameter, vs time (x-axis). The x-axis scale is in weeks; the test results here are for about a half-year.
The top graph (part D) shows the current. The middle graph (part G) shows the voltage. The bottom graph (part J) shows the light intensity.
Observations...
* As judged by both current and voltage, the device worked.
* More specifically, the voltage was more than enough to power the system. The required voltage is shown by the dashed line, labeled "CPU threshold", near the bottom of Part G. You can see that the delivered voltage was always above that. (Also... the system was programmed so that if the voltage fell below the requirement, it would have switched to using electricity from the USB port -- and would have remained that way. You can see that the voltage never switched to the USB voltage.)
* There are continual short-term fluctuations, most evident in the current (and clearer in the graphs with an expanded time scale). Why? Day vs night. The device is powered by sunlight. But photosynthetic organisms metabolize at night, using the sugar they have made during the day. It works, in the present context. (There is no addition of a source of energy other than light. Artificial lighting might have played a role in this test, but other tests, in the lab, were done with controlled light-dark cycles.)
The half-year of results reported here is for the time prior to submitting the article. News reports note that the device continued to operate for another six months (and may still be operating).
The "zoom" labels refer to other parts of the full figure, showing expanded data over a short period.
This is part of Figure 2 from the article.
|
It's proof of concept. A microprocessor running on bio-electricity, more specifically on solar power captured by photosynthesis. It's simple, and apparently rather robust. It could be suitable for remote applications requiring low power for extended periods. Whether it will find a place on your window sill is open for now.
One complexity we haven't mentioned yet... What is growing in that chamber? They inoculated it with a cyanobacterial culture. But analysis at later times showed a complex mixture of bacteria. They did not attempt to maintain sterility, but the important question is, do these other bacteria matter? It may be, for example, that other bacteria are important for establishing good contact with the aluminum electrode. Perhaps other well-chosen strains should be added at the start. Related, we also note that the scientists are not sure how the device actually works, and it is possible that varies day vs night, and over time. These are issues that should be dealt with in further work.
How about the question in the title? Yes, they watered the device. as needed -- 2-3 mL every 7-10 days (p 21 of the Supplement).
And the solar cell alternative? That's open to further analysis, but the current device is made of simple non-toxic materials, and continues to operate in the dark without requiring any added storage device.
The photosynthetic organism used here is a Synechocystis. It is a prokaryote, specifically a cyanobacterium. The news stories, including the press release from the university, consistently use out-of-date terminology, referring to it as an alga, or blue-green alga. The article itself refers to the organism both as bacterium and alga, probably reflecting just casual usage by non-biologists. The incorrect terminology is common, but it is somewhat surprising to see it in materials from the university.
News stories:
* Scientists power a computer with algae and sunlight -- Photosynthesis could be an energy source for the Internet of Things. (Peter Judge, DCD (Data Centre Dynamics), May 18, 2022.)
* Algae-powered computing. (Nanowerk News (University of Cambridge), May 12, 2022.)
* Photosynthesis used to power a microprocessor for over six months. (Ellis Wilde, Chemistry World, May 12, 2022.)
The article: Powering a microprocessor by photosynthesis. (P Bombelli et al, Energy & Environmental Science 15:2529, June 2022.) The Electronic Supplementary Material seems to be freely available at that link, regardless of subscription access to the article.
A recent post about making a small amount of electricity... A battery made of paper, and activated by a drop of water (August 6, 2022).
If you can make a computer use photosynthesis by giving it a cyanobacterium, can you do that with a rat? If an injured heart is short of oxygen, should you try photosynthesis? (June 25, 2017).
A recent post about an unusual cyanobacterium: A primitive cyanobacterium (October 25, 2021).
There is more about energy issues on my page Internet Resources for Organic and Biochemistry under Energy resources. It includes a list of some related Musings posts.
Self-boosting vaccines?
August 26, 2022
Many vaccines are given as multiple doses. For example, the primary series for the COVID mRNA vaccines is two shots about a month apart. What if you could get one shot, with part of it becoming active only a month later?
A new article reports progress toward that goal.
Here are some results for an example, using a 10-day activation.
Part E (left) shows images of three core-shell microparticles over 10 days under physiological conditions. All three microparticles showed about the same behavior. They were substantially intact after 7 days, but the cap came off by day 10, allowing release of whatever might be in the inner compartment (the "core").
The scientists measured the porosity of the microparticles. Part F (right) shows the results. Porosity was low for 7 days, then increased dramatically by day 10.
The scale bars in part E are 100 micrometers. They are at the lower right of each image, and are the same for all.
This is part of Figure 1 from the article.
|
What if you don't want a 10-day activation? Just tune the cap so it comes off at the desired time. For example...

|
This graph shows the kinetics of release from microparticles with three different cap materials. For each, release occurs over a narrow time range: at about 9, 25 or 33 days. The last of those is in the range desired for the two primary COVID shots.
This is Figure 3E from the article.
|
What are these cap materials? The cap is based on PLGA (a copolymer of lactic and glycolic acids), a material already approved for medical use. The three variations shown in the figure above differ in molecular weight and/or the end group of the polymer chain. These properties were more important than other properties studied, including particle size and shape.
Could we get microparticles with longer delays? In other work, they have shown microparticles that release at specific times out to a few months.
It's a promising development. The team has already formulated a polio vaccine that is now being tested in lab animals.
News stories:
* Microparticle Vaccine Provides Boosters Automatically. (Medgadget, July 27, 2022. Now archived.)
* 'Self-Boosting' Vaccines Could Be Immunizations of the Future. (Julie Stewart, WebMD, August 4, 2022. Now archived.)
* Microparticles could be used to deliver 'self-boosting' vaccines. (Nanowerk News (Anne Trafton, MIT), July 15, 2022.)
The article, which is open access: Experimental and computational understanding of pulsatile release mechanism from biodegradable core-shell microparticles. (Morteza Sarmadi et al, Science Advances 8:eabn5315, July 13, 2022.)
More about delivering things to the body: A robust capsule for providing micronutrients (January 26, 2020). From the same lab, that of Bob Langer at MIT. Langer is a chemical engineer well known for clever and useful ideas.
A recent post on alternative vaccine technologies: A gastric auto-injector, which gives shots in the stomach lining (January 24, 2022).
This post is listed on my page Biotechnology in the News (BITN) -- Other topics in the section Vaccines (general). The section has a list of Musings posts on various vaccine issues.
August 24, 2022
Briefly noted... A record short day
August 23, 2022
June 29, 2022, was a short day -- 1.59 milliseconds less than 24 hours. It was the shortest day recorded in the modern era, since the advent of atomic clocks. Day length varies for many reasons, on various time scales. The Moon and the atmosphere affect Earth's rotation, as possibly do movements inside the Earth. It is understood that the Earth spun much faster, leading to much shorter days, long long ago. But now the variations from day to day are in the millisecond range. The Earth has been speeding up recently, leading to a series of record-short days in recent years. Exactly why is not clear. The trend toward shorter days could lead to the need for a negative leap second. (One millisecond per day is about 1/3 of a second per year.)
* News story: Earth Sets New Record for Shortest Day -- Earth keeps spinning faster - but why? timeanddate checks the latest numbers. (Graham Jones & Konstantin Bikos, timeanddate, July 27, 2022.)
* More about day length:
- Effect of climate change on timekeeping (August 14, 2024).
- Did changes in Earth's rotation promote the rise of oxygen-evolving photosynthesis? (August 23, 2021).
- Chile earthquake caused the day to become shorter (March 8, 2010).
A tower of (solar) power -- which makes kerosene
August 22, 2022
Solar-powered airplanes? It's a bit tricky, though there are some experimental examples.
But we might use the solar energy to make airplane fuel. Kerosene, for example. And we might do that in an integrated process.
That's the goal of work described in a recent article.
The first figure shows the idea -- and the actual pilot plant...
Part a (left) shows the plan:
- An array of reflectors beam the solar energy to the top of the tower.
- In the tower, water and carbon dioxide, H2O + CO2, are reacted to make hydrogen and carbon monoxide, H2 + CO, a mixture called synthesis gas, or "syngas". That is a common industrial product, made here driven by solar energy. The process was tuned to make a specific mixture optimized for the following steps.
- The syngas is used to make hydrocarbon fuel, approximating a mixture of kerosene and diesel. This is done in a separate building, called the gas-to-liquid (GtL) unit.
Part b (right) shows their pilot plant, which was used for the experimental work reported in the article. (The GtL unit is not visible here.)
This is Figure 1 from the article.
|
Does it work?
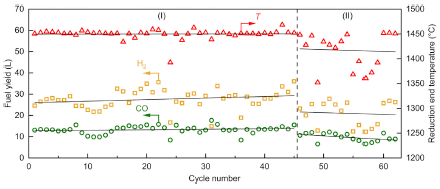
One important feature of a big industrial process is that it works repeatedly. The following figure summarizes some results over a series of runs...
The graph shows some results for the syngas production part of the system. It was run for 62 cycles over a nine day period.
The lower two lines show the amounts of the two product gases: H2 (yellow) and CO (green); see the left-hand y-axis scale. The upper line shows the operating temperature (T) at one step; right-hand y-axis scale.
You can see that the process ran fairly reproducibly for about 45 cycles.
This is Figure 3 from the article.
|
Let's refine the previous question... How well does it work?
It's a mixed bag.
On the plus side, they have shown that the multi-step process does work -- not just at lab scale but in a small (pilot) plant.
On the other hand...
* The attempt to run for an extended period, shown above, had limited success.
* The current process operates at about 4% efficiency. (That is for the production of syngas.) The authors say that 15-20% efficiency would be necessary to make it economical.
They know several ways to improve the efficiency. Some are just a matter of installing known technology, for example, for recovery of waste heat.
Overall, they think an economical process can be established -- but they have not done that yet.
In principle, the use of kerosene from this process would be carbon-neutral. The CO2 from burning the kerosene would merely replace the CO2 that was used to make the kerosene. They did not use CO2 directly from the air in this work, though they have done so at a smaller scale. (The article is not very clear on this point, and some of the news stories may be wrong.)
Progress, but not success.
News stories:
* Solar jet fuel production from CO2 and water scaled up in field demo. (Anthony King, Chemistry World, July 26, 2022.)
* This Solar Tower Can Transform Water, Sunlight, and Carbon Dioxide Into Jet Fuel -- One plant could collect 100 MW of solar radiative power to produce about nine million gallons of kerosene per year. (Tim Newcomb, Popular Mechanics, August 9, 2022.)
* All-in-one solar-powered tower makes carbon-neutral jet fuel. (Science Daily (Cell Press), July 20, 2022.)
* How "Hot Solar" Aviation Fuel from H2O and CO2 Has Taken Off. (Susan Kraemer, SolarPACES, August 10, 2022.) Includes an interview with senior author Aldo Steinfeld.
The article, which is open access: A solar tower fuel plant for the thermochemical production of kerosene from H2O and CO2. (Stefan Zoller et al, Joule 6:1606, July 20, 2022.)
More syngas: Flash photo-pyrolysis: converting banana peels to useful chemicals (March 5, 2022).
More jet fuel: Engineering E coli bacteria to convert cellulose to biofuel (December 13, 2011).
There is more about energy issues on my page Internet Resources for Organic and Biochemistry under Energy resources. It includes a list of some related Musings posts.
How oocytes limit oxygen-induced aging
August 20, 2022
Female humans make their egg cells early, and store them, in a premature form, for decades. How do those cells survive for so long? One might expect routine damage from oxygen to kill them long before the time they are commonly used.
A new article offers some clues.
Let's start with an overview of the situation, including the main finding of the current article...
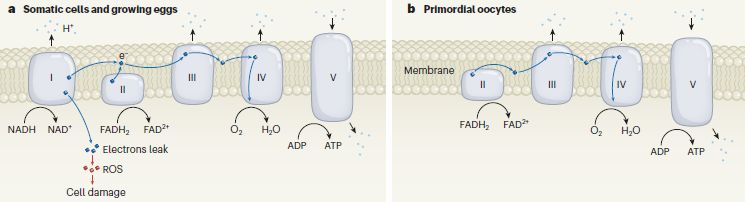
Part a (left) diagrams the usual electron transport ("respiratory") chain that is embedded in the mitochondrial membrane. Briefly, for our purposes...
NADH is oxidized at Complex I. FADH2 is oxidized at Complex II. Both yield electrons, which progress through the chain. The electron paths are shown by wavy lines; most of these are near-horizontal and within the membrane.
Complex IV reduces O2, using those electrons.
Most of the oxidation steps release H+ (protons) into the mitochondrial matrix (inside the organelle; shown here above the membrane). These are shown by dots at the top; note the upward arrows. These H+ are used by Complex V to make ATP; note the downward arrow.
Part b (right) shows the electron transport chain as found in "primordial oocytes" -- early stage egg cells. It is similar, with one important difference: Complex I is absent.
And that leads to one more point... The electron transport chain can leak electrons, producing ROS (reactive oxygen species), which can damage the cell. It turns out that most of the electron leakage comes at Complex I; see the labeling just below Complex I at the left.
The primordial oocytes at the right lack Complex I, and thus have little electron leakage -- and little ROS. That is what the current article is about.
The most familiar ROS is probably peroxide, but the most important may be the superoxide ion, O2-.
This is Figure 1 from the News story accompanying the article (Adhikari & Carroll).
|
The article provides evidence for these points, and suggests that the absence of Complex I is why oocytes can survive for decades with little damage.
The scientists worked with oocytes from humans and from the frog Xenopus laevis, a common lab model organism. Interestingly, their oocyte stories seem to be similar.
Here are a couple of the experiments from the article...

|
This experiment measured the ROS from early-stage oocytes and from the surrounding cells, called granulosa cells. These are shown as the left and right bars, respectively. Part b (top) is for human; part c (bottom) is for Xenopus.
For both animals, the level of ROS was quite low for the oocytes.
The measurement used a dye which changes color upon reaction with ROS.
This is part of Figure 1 from the article.
|
The experiment above helps to establish that ROS is low in primordial oocytes. The next experiment provides evidence for the lack of functional Complex I in those early oocytes.
The top half of the figure shows a measurement of Complex I activity in Stage I (early) and Stage VI (late) Xenopus oocytes.
Start at the right, with the late oocytes. The green curve shows the progress of the reaction over time. The top curve (black) shows the effect of an inhibitor known to act at this step. It works.
The left side shows the same test for the early oocytes. No activity (and the inhibitor has no effect).
The bottom half of the figure is a control. It shows a similar test for another complex of the electron transport chain (IV). The results are similar for both early and late oocytes.
The x-axis presumably shows assay time, probably in minutes. But the graph and legend don't say so.
This is part of Figure 4b from the article. A final frame of the full Figure 4b shows the results for muscle cells; they are similar to the results for late oocytes.
|

|
A picture emerges: oocytes avoid oxidative damage for decades because they avoid the step that generates ROS. The story is more complete for Xenopus, but seems to hold for humans as far as they have looked.
The article discusses a range of fertility issues, some of which may relate to ROS. It is known that women vary in how their fertility declines with age. A better understanding of oxygen-induced damage, building on the current work, could have implications for fertility.
News stories:
* Egg cells remain dormant for decades by putting mitochondria to sleep. (Eleanor Gallegos, IVF.net, August 4, 2022.) The page is from BioNews, which is now being distributed by Progress Educational Trust.
* Egg Cells Maintain Reproductive Longevity by Switching to "Standby Mode". (Sarah Whelan, Technology Networks, July 20, 2022.)
* Human eggs remain healthy for decades by putting 'batteries on standby mode'. (CRG (Center for Genomic Regulation, Barcelona), July 20, 2022.) From the lead institution. The page contains the full press release in English, Spanish and Catalan.
* News story accompanying the article: Developmental biology: A self-defence strategy for long-lived eggs -- Egg cells need to stay out of harm's way to keep the next generation healthy and free of unwanted mutations. A mechanism by which eggs avoid the ravages caused by harmful reactive oxygen species has now been discovered. (Deepak Adhikari & John Carroll, Nature 607:664, July 28, 2022.)
* The article, which is open access: Oocytes maintain ROS-free mitochondrial metabolism by suppressing complex I. (Aida Rodríguez-Nuevo et al, Nature 607:756, July 28, 2022.)
Post about ROS include:
* Tau and ALS (February 19, 2022).
* Coral bleaching: how some symbionts prevent it (September 30, 2016).
* Anti-oxidants and cancer? (October 18, 2015).
A post about oocyte aging: Increased risk of congenital heart defects in offspring from older mothers: Why? and can we do anything about it? (July 18, 2015).
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Aging.
August 17, 2022
Briefly noted... Are venoms sterile?
August 17, 2022
The medical community has generally assumed that venoms are sterile. A recent article shows that is wrong. In fact, they may contain unusual microbes that are hard to treat. An exotic story, but it may be important as well as interesting. For example, bite-associated infections are common; they are often considered secondary infections, but may well actually be due to the bite.
* News story: Bacteria Can Live in Venoms of Snakes and Spiders, New Study Says. (Enrico de Lazaro, Sci.News, May 24, 2022.)
* The article, which is open access: Bacterial Adaptation to Venom in Snakes and Arachnida. (Elham Esmaeilishirazifard et al, Microbiology Spectrum 10:02408-21, May 2022.)
* Also see: Snake venom gland organoids (March 17, 2020). Links to more.
Purifying water using fluorinated nanopores
August 16, 2022
Desalination is expensive. It requires separating the water from a soluble salt -- by pushing the water through a membrane. That takes a lot of energy.
Perhaps there is a hint as to how we might do better in the kitchen. Things don't stick to Teflon, a fluorinated polymer. Further, there are proteins whose job it is to transport water across membranes -- and it is now known that the pores are quite hydrophobic. (Such proteins are called aquaporins.)
So, can we make better membranes for purifying water by making the pores hydrophobic? A recent article addresses the question, and offers some encouraging results.

|
The top part of the figure shows some of the chemistry specifics. The bottom part is a cartoon diagram of the idea.
The chemical structure at the top is one ring molecule the scientists made. It has a hole in it -- 1.76 nanometers diameter in this case. (They made several such chemicals, with different hole sizes. The full part A in the article shows them.) The green region in the structure shows lots of fluorine atoms. A stack of such ring molecules in a membrane serves as the functional pore -- an F-lined hydrophobic pore.
Part C (bottom) shows a hydrophobic pore at the right, and a less-hydrophobic pore at the left. Each blue dot is a water molecule. The water molecules tend to form clusters, impeding their flow through narrow channels. But the water molecules have no attraction to the more hydrophobic surface; that disrupts the clusters -- and might lead to faster flow.
This is part of Figure 1 from the article.
|
Does it work? Here is a test...
The graph shows the water flow through various materials. The flow is water volume per second per membrane pore area, cm3 s-1 nm-2. The four bars you can see are for four of the F-lined pores, such as the one shown above.
The two materials at the right gave such low flows that there is no bar. However, the numeric value is shown. The new F-lined pores are all about 100-fold better than these two materials at the right -- carbon nanotubes (CNT) and a natural water-carrying protein (the aquaporin AQP1).
This is Figure 4B from the article.
|

|
The results support the idea that using such hydrophobic pores could promote water flow. The material with the best performance here is the one with the smallest pores, about 0.9 nm.
Promoting water flow is only half the battle. Water purification membranes must also reject the solutes, in particular salts. One would expect these hydrophobic pores to reject the ions. The scientists do one test, which shows that is true.
These membranes with F-lined pores are hard to make. It is unlikely that they would be practical, at least as made here. But they provide a lead for further development.
News stories:
* Japan Develops Fluorine-Based Nanomembrane for Quick, Efficient Water Desalination. (Ron Jefferson, Science Times, May 16, 2022.)
* The future of desalination? -- A fast, efficient, selective membrane for purifying saltwater. (EurekAlert! (University of Tokyo), May 12, 2022.)
* Ultrafast permeation of water through nanochannels with dense fluoride inner surfaces. (iNEWS, undated.) A lengthy and quite technical presentation.
* News story accompanying the article: Materials science: Beating natural proteins at filtering water -- Artificial fluorous channels outperform aquaporins in water permeation. (Yuexiao Shen, Science 376:698, May 13, 2022.)
* The article: Ultrafast water permeation through nanochannels with a densely fluorous interior surface. (Yoshimitsu Itoh et al, Science 376:738, May 13, 2022.)
Posts about desalination include:
* Better membranes for water desalination (February 14, 2021).
* Water desalination using graphene oxide membranes? (April 29, 2017).
More on water purification: Briefly noted... Recycled water: safety (May 24, 2023).
More on development of membranes for filtration: Fractionating crude oil using a membrane (September 2, 2022).
Among posts about the effects of a Teflon coating...
* Miller-Urey revisited: the role of the glass container (January 22, 2022).
* An antiviral coating for medical textiles (July 12, 2020).
A phage treatment for inflammatory bowel disease
August 13, 2022
A new article offers a treatment for inflammatory bowel disease (IBD). The work was based on analysis of several hundred patients, with various kinds of IBD from various regions around the world. Here are some pieces of the story.
The first figure provides evidence that a particular kind of bacteria is associated with IBD...

|
Each row shows the amount of a certain type of bacteria during disease flare-ups and during remission ("Rem"). The key for this heat map is shown at the lower right, but it is typical. Red is high, blue is low.
One of the strongest signals is in the top row. That is for the bacteria Klebsiella pneumoniae.
This is part of Figure 1D from the article. The list of bacteria is at the far left of the full figure.
|
Those results suggest an association between IBD and Klebsiella pneumoniae (Kp) bacteria. Results such as these, by themselves, do not show a causal connection, but it is known that Kp can be inflammatory, and there is work on this point in the article.
The scientists decided to target Kp -- using bacteriophages.
That was actually a major project in itself. Not all phages work well. At some point, they came up with six candidate mixtures, each with five phages. These mixtures are labeled 5(A) through 5(F).
The following figure shows a test of how these phage mixtures affect growth of Kp bacteria. The test is in lab cultures of the bacteria.
The graph shows optical density (600 nm), a measure of bacterial growth, vs time.
The top curve is the control, labeled "Veh" for vehicle; normal growth. The bottom curve, at OD = 0, is for a "blank" with no bacteria.
The six main curves fall into two general groups. Two of the phage mixtures gave curves very near the bottom, showing that they did a good job of stopping the bacteria, even over 20 hours. Four other mixtures did not do as well, even with five phages.
This is part of Figure 5D from the article.
|

|
So we now have a candidate bacterial cause and a candidate treatment. How about a test...
The test here is a model system with mice. The mice were infected with Kp bacteria (same strain as above), and given a good 5-phage mixture as a treatment.
The two graphs show the amount of two mouse proteins considered markers of inflammation. Each point shows the level of that protein in one mouse. The data to the left in each graph is for the control ("Veh"); the data to the right is for the phage treatment.
In both cases, there is a general trend toward having less of the inflammatory protein upon phage treatment.
This is part of Figure 6 from the article.
|

|
It's encouraging. A treatment targeted to specific bacteria that cause inflammation.
The authors did one test with humans; healthy volunteers tolerated the phages well, and the phages themselves survived. Further testing of the phage treatment in humans with IBD is planned.
IBD is heterogeneous. (Ulcerative colitis and Crohn's disease are sub-types of IBD.) One would not expect a single phage mixture to treat all cases. The immediate practical question is whether what the scientists have done here carries over to some people.
News stories:
* Scientists Identify Viruses as New Weapons to Fight IBD. (Maya Davis, WebMD, August 9, 2022. Now archived.)
* Phage combination therapy can precisely target IBD-related gut bacteria without harming helpful microbes. (Science Daily (Cell Press), August 4, 2022.)
The article: Targeted suppression of human IBD-associated gut microbiota commensals by phage consortia for treatment of intestinal inflammation. (Sara Federici et al, Cell 185:2879, August 4, 2022.)
An earlier post about phage therapy: A virus that could treat acne? (October 21, 2012)
Posts relating to IBD include...
* Treating asthma with a hookworm protein? (December 2, 2016).
* Is Arthromitus a key bug in your gut? (January 16, 2010).
An earlier post about Klebsiella: Melamine toxicity: possible role of gut microbiota (April 21, 2013).
There is more about phage therapy on my page Internet resources: Biology - Miscellaneous in the section Medicine/microbiology: phage therapy.
Update November 9, 2023...
A major phage therapy project has reported five years of systematic clinical experience. The work focused on people with infections where conventional treatment was unsuccessful. The phage therapy was then administered on a compassionate-use basis, not in controlled trials. The results are encouraging, with about 80% success. That success rate is based on the number of patients actually treated, not the number considered. There are two articles, both from 2023. #1 is general, involving various types of infections. #2 focuses on Pseudomonas, using a single phage.
* Article 1:
- News story: Israeli researchers report high success rate for compassionate-use phage therapy. (Chris Dall, CIDRAP, April 24, 2023. First item on the page.)
- The article, which is open access: Compassionate Use of Bacteriophages for Failed Persistent Infections During the First 5 Years of the Israeli Phage Therapy Center. (Hadil Onallah et al, Open Forum Infectious Diseases 10:ofad221, May 2023.)
* Article 2:
- News story: Phage therapy shows promise in treating tough Pseudomonas aeruginosa infections. (News-Medical.net (Hebrew University of Jerusalem), August 7, 2023.)
- The article: Refractory Pseudomonas aeruginosa infections treated with phage PASA16: A compassionate use case series. (Hadil Onallah et al, Med 4:600, September 8, 2023.)
August 10, 2022
Making decoys that trap the SARS-2 virus
August 10, 2022
The idea of decoys is straightforward. Practical implementation of a therapeutic decoy has not yet been achieved.
A recent article explores making decoys for the SARS-2 (COVID-19) virus. In cartoon form...

|
The irregular large object at the left is a cell.
The Y-shaped things attached to it are virus receptors.
It is a natural process for cells to bud off small vesicles. They have normal cell membranes on the surface. In this case, that includes the virus receptors. The figure shows three such vesicles just to the right of the main cell; each has six virus receptors. (The inset shows one vesicle enlarged.)
|
If virus absorbs to the receptors on these vesicles, there will not be a productive infection; the virus is effectively dead. The vesicles have served as decoys to prevent infection.
Why doesn't the virus grow in the vesicles? The vesicles are small, and do not contain a full complement of cellular resources.
There is no significance to the number or position of receptors shown in the diagrams.
This is part of Figure 1 from the article. As the arrow (lower right) suggests, there is more to the figure. But a caution... I found this and some other figures in the article to be confusing.
|
The current work is about making decoys for SARS-2. The cells are a human cell line, one that is good at making vesicles. The cell line has been modified so that it makes a large number of receptors for the SARS-2 virus (a protein called ACE2 = angiotensin-converting enzyme 2).
The scientists collect and purify the vesicles.
The following figure shows some results testing how well the vesicles work as decoys, in a simple lab test.

The graph shows percent infections (y-axis) vs dose of vesicles added (x-axis, log scale). EV = extracellular vesicles.
The top two curves (black, gray) are for vesicles without viral receptors; they are labeled "ACE2 negative". These two curves gave near 100% infections across the entire range of doses, as expected.
The other four curves are all for various vesicles with viral receptors. All four reduced the number of infections, in a dose-dependent manner. Good; that's the idea.
What is different about them? One pair of curves is labeled "WT ACE2"; that stands for the wild type receptor. The other pair is labeled "Mut ACE2"; it used a mutant receptor that binds the virus more tightly. Indeed, it took less of these mutant-receptor vesicles to reduce the infections.
The curves come in pairs. The two members of each pair are labeled HS and UC. Those are for different ways of purifying the vesicles. The HS vesicles are a bit more effective, perhaps due to being slightly larger and carrying more receptors.
The viral infections here use a lab trick. You don't need to understand it to follow the graph, but the procedure is interesting, and common. The "virus" is an artificial construct. Key to its use here, it carries the SARS-2 Spike protein, so it attaches to cells (or decoys) just like the SARS-2 virus does. Upon infection, it does little except to cause production of an easily measured protein. That is, it is a safe particle that does what is needed here.
This is Figure 3D from the article.
|
Well, that is a start. The general effectiveness of a variety of decoys is more important than small differences between them. We'll see whether the scientists can turn this into a useful drug. They note that no decoy treatment has yet gone to human clinical trials.
A concern about any treatment is the possible development of resistance. The authors make an interesting argument for their decoys. The decoys themselves are actually playing a normal biological role: binding the virus, in a normal manner. If a virus develops that infects better because it uses the receptor better, it might reasonably also use the decoy better. They provide a little evidence to support this for a small set of variants. There is no assurance that this will always hold.
News stories:
* Decoy particles trick SARS-CoV-2. (Victoria Corless, Advanced Science News, April 22, 2022.)
* Decoy nanoparticles trick coronavirus as it evolves. (Nanowerk News (Amanda Morris, Northwestern University), April 12, 2022.)
The article, which is open access: Elucidating Design Principles for Engineering Cell-Derived Vesicles to Inhibit SARS-CoV-2 Infection. (Taylor F Gunnels et al, Small 18:2200125, May 12, 2022.)
Post about decoys include...
* Briefly noted... Decoy receptors. (November 4, 2020). A natural process in mammalian cells.
* Why don't black African mosquitoes bite humans? (December 19, 2014).
* A modified chicken that cannot transmit bird flu (March 26, 2011)
There is a BITN section for SARS, MERS (coronaviruses). It includes a list of Musings posts in the field.
Briefly noted... Legionella bacteria and the origin of eukaryotes
August 9, 2022
A recent article reports analysis of numerous genomes from the bacterial order Legionellales, which contains the familiar genus Legionella, suggests that the group originated nearly two billion years ago, very near the time of the origin of eukaryotic cells. Since all Legionellales gain entry to their eukaryotic host cell by phagocytosis, this suggests that phagocytosis was present in eukaryotic cells two billion years ago. That may well be before the origin of mitochondria. The article offers some intriguing perspective on the early development of eukaryotic cells. But a big caution... The dates have huge uncertainties; the suggestions about the order of events are not very solid.
* News story: Ancestors of Legionella Bacteria - Which Causes Legionnaires' Disease - Infected Cells Two Billion Years Ago. (SciTechDaily (Uppsala University), February 15, 2022.)
* The article, which is open access: Host Adaptation in Legionellales Is 1.9 Ga, Coincident with Eukaryogenesis. (Eric Hugoson et al, Molecular Biology and Evolution 39:msac037, March 2022.)
* There are many posts about the origin of eukaryotes. Here is one that mentions the phagocytosis issue: An Asgard in culture (February 4, 2020).
* Previous post about Legionella: Can you get sick from the street cleaning truck? (December 10, 2017).
Do Vitamin D supplements prevent bone fractures?
August 8, 2022
Vitamin D is important -- and is still the subject of much discussion. We know, for example, that vitamin D is involved in formation of bones. However, the question of optimal dose is much harder to establish. That leads to the question of whether taking supplemental vitamin D would be useful.
A new article reports results from a clinical trial addressing whether vitamin D supplements reduce the incidence of broken bones in middle-age or older adults.
Here is the summary...
The graph shows the incidence of bone fracture in the two groups: those given a vitamin D supplement vs the control group, given a placebo. The incidence is plotted vs time of follow-up in the trial.
The main graph shows no effect.
The inset shows the same results with an expanded y-axis scale. There is no effect, as confirmed by statistical analysis.
This is Figure 2A from the article.
|
That's the big picture. One can look at some details, such as various types of fractures. Such analyses are shown in the following table...
In the table, the right-hand column is hazard ratio (HR), with 95% confidence limits. A HR of 1.05 would mean that the vitamin D supplement increased the risk of fracture by 5%; a HR of 0.95 would be for a 5% decrease.
Careful, the numbers are small. The numbers in parentheses are the 95% confidence limits of the HR value; all confidence limits shown include 1.0.
The top row of the table is for the results shown in the first figure.
This is Table 2 from the article. Table 3 shows analyses by sub-groups of the trial participants. Again, no statistically significant effects were found.
|
Musings has noted an earlier article about the same trial [link at the end]. In that post, we noted some of the limitations of the trial. Those comments hold here, too. In particular, the trial included people without regard to their vitamin D or bone-health status, so long as they were generally healthy.
In the current work, the results were analyzed to look for an effect of the measured vitamin D levels. No such effect was found. However, there were few people with very low levels, making the test of limited value.
Results of various trials of vitamin D over the years have been inconsistent. It seems likely that we are still missing basic information about bone health.
News stories:
* Study Finds Vitamin D Supplements Do Not Reduce Risk of Broken Bones. (SciTechDaily (Brigham and Women's Hospital), July 30, 2022.)
* Vitamin D Supplement Does Not Reduce Fracture Risk in Older Adults. (Physician's Weekly (HealthDay News), July 28, 2022.)
* Editorial accompanying the article: VITAL Findings - A Decisive Verdict on Vitamin D Supplementation. (Steven R Cummings & Clifford Rosen, New England Journal of Medicine 387:368, July 28, 2022.) A broad view of the trial results, including this article.
* The article: Supplemental Vitamin D and Incident Fractures in Midlife and Older Adults. (Meryl S LeBoff et al, New England Journal of Medicine 387:299, July 28, 2022.)
Background post about the same trial: Vitamin D, omega-3 fatty acids, and autoimmune disease? (March 29, 2022).
More Vitamin D:
* Briefly noted... Vitamin D and COVID-19? (June 23, 2021).
* Vitamin D: How much is too much? (July 9, 2013).
My page Internet resources: Biology - Miscellaneous contains a section on Nutrition; food and drug safety.
A battery made of paper, and activated by a drop of water
August 6, 2022
Need a little electricity? Just take out a little battery, and add a drop of water to activate it. Toss when done.
That's the gist of a new article, exploring one battery niche.

You can consider the two parts of this figure in either order.
I'll start with part c (bottom), for fun. It shows the new battery in use. The use here is a small clock, with a liquid crystal display. The battery drives the display, as well as the operation of the clock.
The battery itself is shown to the left, complete with a bit of advertising for the institute doing the work. The lettering is actually the active part of the battery, which can be produced in whatever shape is desired.
Part a (top) gives an idea of what is in the battery. The dry battery (left; white background) has some zinc in it -- and is inactive. Add a drop of water (right; blue background), and some aqueous chemistry starts -- electron transfer reactions that produce electricity.
The zinc dissolves, as Zn2+ ions, with two electrons released as electricity; see the bottom circle of reactions. The circles show the reactions at various sites. The electrons ultimately go to O2; see the top circle.
The addition of water dissolves NaCl that was also embedded in the paper. That creates an electrolyte solution. That effectively activates the battery.
The clock runs until the battery runs dry -- literally, in this case. See the next figure.
The battery for the clock has two cells in series. You can (barely) see a vertical line between the m and p, it is a bit of wax, as a hydrophobic boundary between cells.
This is part of Figure 1 from the article.
|
The following figure shows some data for the electrical performance of the battery...
The graph shows voltage vs time for a single-cell battery. In this case, the battery was operated at constant current (100 µA).
The battery gave about 0.5 V when fresh. It stopped working at about 1 h. Adding a bit more water restored its performance -- for another hour or so.
The break in the graph at 0.5 h was due to the scientists interrupting the work to take other measurements.
This is Figure 3f from the article.
|
The capacity of the battery is determined by the amount of zinc it contains. Batteries for a particular application can be manufactured with the right amount of Zn, to minimize metallic waste.
This is not just about making a small battery, but a simple one. Simple to make, simple to use, simple to dispose.
News stories:
* Disposable paper battery is activated by just a drop of water -- Tiny, cheap batteries like this could someday turn anything into an electronic device. (Tibi Puiu, ZME Science, July 29, 2022.)
* A paper battery with water switch. (Nanowerk News (Empa), July 29, 2022.)
Empa? It's an acronym based on the German name of the institute, which you can find on their Wikipedia page. But even with that name at hand, it's a bit of a stretch.
The article, which is open access: Water activated disposable paper battery. (Alexandre Poulin et al, Scientific Reports 12:11919, July 28, 2022.) A very readable article.
Previous post about batteries: A better membrane for vanadium-based flow batteries (March 11, 2022). Big batteries, here.
Another Zn-based battery: Boiled batteries (July 19, 2010). Batteries using Zn are common.
Also see: If your computer was powered by photosynthesis, would you have to water it? (August 29, 2022).
There is more about energy on my page Internet Resources for Organic and Biochemistry under Energy resources. It includes a list of some related Musings posts.
August 3, 2022
Briefly noted... Faked data and the status of research on Alzheimer's disease
August 3, 2022
There is a stir in the field of Alzheimer's disease (AD) research. A news story in Science in late July brought to the public a concern about possible data manipulation in an important paper from 2006 (and follow-up papers). That leads to the question of how research in the field was affected by an (apparently) invalid article. Scientific misconduct is an important issue; the charges will be the subject of appropriate scrutiny. But AD research is itself an important topic, and the current incident is a chance to try to get a big picture view. It is something of a mess.
* News story: Faked Beta-Amyloid Data. What Does It Mean? (Derek Lowe, In the Pipeline (AAAS), July 25, 2022.) Links to the original news story in Science. That story is good for describing some of the background of the data problem, but Lowe's story focuses on the science of AD.
* This is listed on my BITN -- Other topics page in the section Alzheimer's disease. (I don't see any Musings posts about the suspect work, at least directly. The original article in question pre-dates Musings.)
What if infection triggered a treatment?
August 2, 2022
It's an intriguing idea. How might it work? Imagine that you have an antibiotic "stored" in your body. Bacteria infect you, and an enzyme from the bacteria releases the stored antibiotic.
A recent article works out part of a possible process.
Here's the idea...
Start with the two blue cylinders (just left of center). The upper and lower cylinders are labeled: "responsive hydrogel (R)" and "non-responsive hydrogel (NR)". (Don't worry about the chemical details (at the left) for now.)
Both hydrogels have orange circles in them. Those represent "nanoparticle cargo", and come from the step to the left. That is, "cargo" is incorporated into the gels when they are formed.
To the right of those cylinders... Add the enzyme β-lactamase. The responsive hydrogel responds, and breaks down, releasing the cargo. The non-responsive hydrogel does not respond; it does not break down, and the cargo is not released.
This is Scheme 1 from the article.
|
We'll come back to parts of that below, but first... Does it work? Look...
The figure shows the NR and R hydrogels over time after adding the enzyme.
The responsive (R) hydrogel is degraded. That's quite clear by the 1 hour time point; it is probably visible in the 0.5 h point. The non-responsive (NR) hydrogel remains unaffected over the entire time period.
This is Figure 1a from the article.
|
Here are some quantitative data about such a test...

It's a complex figure. There are four data sets, each with three curves. The data sets show the mass of the hydrogels (y-axis at the left) and nanoparticle (NP) cargo release (y-axis at the right) vs time of enzyme treatment (x-axis). The three curves in each set are for three different particle sizes of NP cargo.
The two data sets that look interesting, in the middle, are for the R hydrogel. The mass decreases, and the cargo is released. For cargo release, the blue curve is a bit lower than the others; it is for the largest size cargo particles (100 nm; the others are 25 and 50 nm).
The non-responsive gel shows no change in mass and no cargo release (top and bottom sets, respectively, more or less just horizontal lines). (There is only one curve in the bottom data set.)
The model cargo used for testing is fluorescent, allowing for easy measurement.
This is slightly modified from Figure 2a of the article. I have added labels; when relevant, the label applies to a group of three lines (for different size NP).
|
The big picture is that they have made a gel that can store cargo and respond to an enzyme by releasing the cargo.
The enzyme used here is β-lactamase, which is produced by some infecting bacteria. In particular, such an enzyme is associated with bacteria resistant to β-lactam antibiotics (e.g., penicillins). The gel is sensitive to this enzyme, because the scientists have built the β-lactam feature into the gel. This chemistry is shown in the top figure at the left. The blue feature of the chemical structure is a β-lactam,
One test was done on pig skin, a common lab model for studying infections. The presence of enzyme-producing bacteria caused cargo release with the R-hydrogel on the skin. That is as far as they got with making the system real biology. There is no real infection, and the cargo is not a real treatment in any of this work. The scientists have shown the first steps in developing a treatment that responds to infection. For example, one might put a hydrogel carrying an antibiotic on a wound, but the antibiotic would be released only if an infection occurred.
News story: New drug delivery system releases therapeutic cargo only when bacteria are present. (Nanowerk News (Kevin Stacey, Brown University), June 8, 2022.)
The article: β-Lactamase-Responsive Hydrogel Drug Delivery Platform for Bacteria-Triggered Cargo Release. (Dahlia Alkekhia et al, ACS Applied Materials & Interfaces 14:27538, June 22, 2022.)
More about hydrogels, enzymes, and potential therapeutics: Engineering a worm to treat cancer (July 23, 2022).
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Antibiotics. It includes a list of Musings posts on the topic.
Briefly noted... James Lovelock, 1919-2022
August 1, 2022
In mid-July I had occasion to note the work of James Lovelock. I also noted that Lovelock would be 103 later in the month. Lovelock died last week, on his 103rd birthday, July 26. Here is one news story; a simple search will turn up much more.
* News story: James Lovelock, creator of Gaia hypothesis, dies on 103rd birthday -- The scientist was best known for his theory that the Earth is a self-regulating community of organisms. (Helena Horton, Guardian, July 27, 2022.)
* I have added this update to that earlier item, Briefly noted... How intelligent is your planet? (July 13, 2022), and to a Musings supplemental page that collects posts about him, Gaia and James Lovelock.
* I have also added this to my BITN - Miscellaneous page, in the section on Aging. That section includes a list of centenarians that we have noted in Musings.
Precipitating CO2 from air
July 30, 2022
One approach to reducing the amount of carbon dioxide in the atmosphere is to capture it. There is much work in the field; it gets subdivided, by the source. It is one thing to capture CO2 from a concentrated source, such as the effluent of an industrial plant that is burring fossil fuel. It is quite another to capture CO2 from the air (direct air capture = DAC). It is a dilute source, and produces a dilute product stream.
A new article reports progress in capturing CO2 from ordinary air.
The following figure shows the chemical that is key to the proposed process, and the capture reaction...
The capture chemical is IPDA (isophorone diamine; the IUPAC name is in the article).
The CO2 reacts with an amino group, forming a carbamic acid, the group at the top of the product chemical, which they call CA1.
This is Scheme 1 from the article.
|

|
Capturing CO2 with amines is common. What's special about using this amine is shown in the next figure...

|
The figure shows the capture system at two time points, Focus on the reaction tube at the right. The bottom of the tube is just above the time label.
The system is to flow gas into the tube. The gas is CO2, at 400 ppm, about the level in the air. Here, it is in pure N2. That's about it. The instruments at the left are for measurements.
The important point is that the product, CA1, is insoluble. It settles to the bottom of the tube, as you can see in the lower photo.
The system not only captures CO2, but collects it in a convenient form.
In this case, the solvent was DMSO, but the system also works with water.
This is part of Figure 3 from the article. (Kinetic data from such an experiment is shown in Figure 2 of the article. It includes comparison with other sorbents.)
|
There is more. In particular, the precipitate is easily processed, at fairly low temperature compared to current processes, to liberate the CO2 and regenerate the original amine. The released CO2, now concentrated, may be useful, or it can be stored.
The process removes 99% of the CO2, and uses the sorbent efficiently (1 mole CO2 per mole IPDA). That is from the gas stream that flows through the device. The efficiency in actual use will depend on how the process is configured. For now, the point is, it works well -- compared to common alternatives.
As a bonus, the reaction is fast, about twice as fast as with other sorbents.
Overall, the article offers an improved way to capture CO2 from air. This is lab-scale work, with no economic analysis.
News stories:
* New Carbon Capture System with Record-Breaking Efficiency to Revolutionise Direct Air Capture -- Newly discovered sorbent is 99 per cent efficient in absorbing low concentrations (400ppm) of carbon dioxide from the atmosphere at unprecedented rates. (Asia Pacific Biotech News, July 2022. Now archived.)
* Fastest carbon dioxide catcher heralds new age for direct air capture. (Nanowerk News (Tokyo Metropolitan University), May 28, 2022.)
The article, which is open access: Direct Air Capture of CO2 Using a Liquid Amine.Solid Carbamic Acid Phase-Separation System Using Diamines Bearing an Aminocyclohexyl Group. (Soichi Kikkawa et al, ACS Environmental Au 2:354, July 20, 2022.)
Among posts on capturing CO2:
* Added September 4, 2025.
Using bacteria to leach valuable metals, including rare earths, from rocks -- and to capture atmospheric carbon (September 4, 2025).
* Capturing carbon dioxide using gallium (April 4, 2022).
* CO2 capture from the air: an improved estimate of the cost (July 16, 2018). The article of this earlier post is reference 40 of the current article.
For the possibility of capturing methane from the air, Harvesting methane from the air? (May 15, 2024).
July 27, 2022
Quantum mechanical tunneling in DNA
July 27, 2022
Information storage in DNA is based on hydrogen bonding. However, the key H atoms can actually shift position, at low frequency, potentially causing mutations.
A recent article explores whether such H-shifts might be due to quantum mechanical tunneling.
The first figure shows the H-shifts being considered here...

The left side shows an ordinary G-C base pair. (R is deoxyribose, and the rest of the DNA chain.)
The right side shows a G*-C* base pair, after two H have shifted.
In the G base, the H labeled "a" moved from the N at the left to the N at the right. That is, this H starts with a covalent bond at its left and a hydrogen bond at its right, and ends up the other way around: a hydrogen bond at its left and a covalent bond at its right. (Confused? Look at the picture.)
In the C base, the H labeled "b" moved from the N at its right to the O at its left.
In each case, the * base has different base-pairing properties than the original base. G* can pair with C* (as shown), but G* can also pair with T -- potentially leading to a mutation if it is involved in replication. Similarly, C* can pair with A.
Chemists call the structures with shifted H atoms tautomers. That is, the G-C and G*-C* base pairs are tautomers. G-C is the major tautomer under ordinary conditions in this case.
This is modified from Figure 1b of article 2. I added the a and b labels on the H atoms that shift. (The corresponding figure in article 1, which is the current focus, is somewhat less clear, by not showing all the bonding detail.)
|
The big idea above -- even if you are confused by the structural details -- is that the bases can shift into alternative forms, which are potentially mutagenic. The question is how such shifts occur. In fact, there are a couple of possibilities.
It could occur by ordinary chemistry. That is not likely; there is a substantial activation energy barrier. But there is another possibility: the double shift could occur by the quantum mechanical process of tunneling.
The current article explores that tunneling process, by theoretical calculations. In particular, the authors use a model that takes temperature into account more completely. Here is what they found...
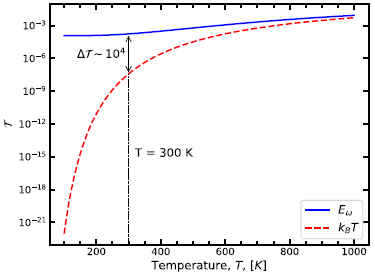
|
Careful. The graph shows Τ on the y-axis, and T on the x-axis. Hard to tell them apart, so stay alert. The y-axis Τ (a capital Tau) is the fraction of the bases in the * form. The x-axis, of course, is temperature.
The upper (solid blue) curve is for quantum tunneling using their more complete model. The lower (dashed red) curve is for a common simpler model.
The vertical dashed line is for T = 300 Kelvin, which is 27 °C. A typical temperature of biology.
|
The picture is clear. At biology temperature, their improved consideration of T in the quantum tunneling shows that the frequency of the tautomers is much higher -- by a factor of 104 -- than previously expected. It is also higher than expected from classical chemistry.
This is Figure 4 from article 1.
|
The improved calculation also shows that the tautomer frequency is high enough that it could easily be a factor in causing mutations. There are more steps to consider, but that is the heart of the current analysis.
In their original paper on the DNA double helix, Watson and Crick suggested that base tautomerism might be a cause of mutations. The matter has been debated over the years; it is a difficult issue, both experimentally and theoretically. The current work uses an improved theoretical framework to make the case that tautomerism is important -- due to quantum mechanical effects.
Another possible tautomerization can occur with free nucleotides. This involves a single H shifting within that base. For example, the H marked "a" in the top figure could shift to the N just above. That is the kind of tautomerization that chemists normally consider, within a single molecule. It can occur in bases in the pool of free nucleotide precursors (such as dGTP). That is distinct from the double-shift event in a base pair, which is the main focus of the current article. Both types of events are potentially mutagenic.
News stories:
* Quantum effects help make DNA unstable. (Isabelle Dumé, Physics World, June 14, 2022.)
* Quantum mechanics could explain why DNA can spontaneously mutate. (Nanowerk News (University of Surrey), May 5, 2022.)
There are two relevant articles, both open access. Article 1, which is recent, is the main focus of the post. Article 2 is related and from the same authors last year; I used their figure for the tautomers.
1) An open quantum systems approach to proton tunnelling in DNA. (Louie Slocombe et al, Communications Physics 5:109, May 5, 2022.)
2) Quantum and classical effects in DNA point mutations: Watson-Crick tautomerism in AT and GC base pairs. (L Slocombe et al, Physical Chemistry Chemical Physics 23:4141, February 21, 2021.)
Also see:
* Making peptides in space: a new pathway (April 15, 2022). The article discussed in this post invoked quantum tunneling at one reaction step, though we did not discuss that in the post.
* The original Watson-Crick paper on the structure of DNA (October 25, 2010).
The senior author of both articles is Jim Al-Khalili. He is the co-author of a book listed on my page Books: Suggestions for general science reading. McFadden & Al-Khalili, Life on the Edge -- The coming of age of quantum biology (2014).
Briefly noted... Relationship between meat consumption and longevity
July 26, 2022
A recent article reports that higher consumption of meat is associated with greater longevity. The analysis is based on United Nations (FAO) data for 175 countries, accounting for 90% of the world's population. The analysis takes into account several possible confounding variables, including caloric intake, urbanization, obesity and education levels. That illustrates the complexity of the story. If there is any big message here, it is probably that the subject is complicated, and our understanding of human nutrition is incomplete. I suggest focusing on the discussion; it is largely about the strengths and weaknesses of various analyses, which reach various conclusions. Use the article as a chance to examine the issues more carefully; don't insist on quick conclusions.
* News story: Study: Meat Consumption is Positively Associated with Life Expectancy. (Enrico de Lazaro, Sci-News.com, February 22, 2022.)
* The article, which is open access: Total Meat Intake is Associated with Life Expectancy: A Cross-Sectional Data Analysis of 175 Contemporary Populations. (Wenpeng You et al, International Journal of General Medicine 15:1833, February 22, 2022.)
* Also see... Plant-based vs animal-based diets (May 7, 2025).
* I have listed this item on my page Internet resources: Biology - Miscellaneous under Nutrition; food and drug safety.
A tunable catalyst
July 25, 2022
Finding the optimum catalyst can be difficult. The theory behind optimizing catalysis is limited, and there is usually much trial and error. What if we could just have one catalyst, and then tune it so it works optimally?
That's the general idea of a recent article.
Here is a diagram...

|
The active catalytic component is the top layer: alumina = aluminum oxide, Al2O3. It is a common catalyst. (Am = amorphous.)
What's special here is that loop at the left. The scientists can apply a voltage to the catalyst -- thus adjusting its electronic properties.
The key is the charge separation between the graphene and silicon layers. (You can see the graphene at the right, where the upper alumina has been removed.)
This is Figure 1a from the article.
|
To test the idea, the scientists carried out a reaction using various voltages on the catalyst. For each voltage, they measured the rate of the reaction over a range of temperatures (T).
The reaction is the dehydration of isopropanol to make propene (propylene) :
CH3CH(OH)CH3 --> CH3CH=CH2 + H2O.
Here are some results...
The bottom curve is for zero (applied) voltage. The optimum temperature (T) is about 115 °C. (The vertical dashed lines mark the optima for the various conditions.)
The other curves are for increasing voltages, from the bottom to the top of the figure, in 1 volt increments.
At the top, with 3 V, the optimum T is about 70 °C -- 45 degrees lower than with no voltage applied to the catalyst.
There are no numbers on the y-axis (rate) scale. The curves are offset, for ease of viewing. The rates are about the same in each case, as you can see from the height of the peak relative to the baseline. What matters is the position of the peak on the x-axis (T) scale.
There is some discrepancy between the vertical dashed lines and what they say is the optimum T. No big deal, but annoying detail.
This is Figure 4a from the article.
|

|
Catalysts speed up reactions by lowering the activation energy. The results above show that applying 3 V across this "catalytic condenser" lowers the optimum temperature for the reaction by about 45 degrees, reflecting a significant reduction in activation energy.
An interesting question is whether this approach could allow for replacing expensive metal catalysts with devices made from inexpensive metals.
News story: Groundbreaking "Chameleon Metal" Invented That Acts Like Many Others. (SciTechDaily (University of Minnesota), May 11, 2022.)
The article, which is open access: Alumina Graphene Catalytic Condenser for Programmable Solid Acids. (Tzia Ming Onn et al, JACS Au 2:1123, May 23, 2022.)
A recent post with perhaps a similar purpose: Briefly noted... A Swiss army knife for catalysis (February 15, 2022).
Other posts about catalyst development include: A simpler way to make styrene (July 10, 2015). Links to more.
Engineering a worm to treat cancer
July 23, 2022
A new article reports progress toward making a worm that can seek out cancer and treat it. It's an interesting story -- quite incomplete at this point.
Let's start with some results -- things they did accomplish.

|
A sample of cells in lab culture was treated. The cells were then stained so that apoptotic (dying) cells showed up as red.
Treated on the right, control on the left.
What was the treatment? A worm. A worm named NAGOX.
|
|
The difference is impressive.
"PI" on the images (upper left) stands for propidium iodide, the stain.
This is Figure 5d from the article.
|
How does the worm kill the cells? It makes hydrogen peroxide.
This graph shows the production of H2O2 by NAGOX and its control, labeled "non-coated".
This is Figure 5c from the article.
|

|
The results above show that the worm can kill cells, and offers evidence that it does so by making H2O2.
The work does not show a preference for cancer cells. The cells used here are cancer cells (HeLa cells), but there is no comparison. In fact, the H2O2 probably kills most any kind of cells at these concentrations.
So why is there a suggestion this might treat cancer? The worm used here is the nematode, Anisakis simplex. Previous work has shown that it is attracted to cancer cells. You can see where this is going. But it is important to note that it hasn't gone there yet.
Why is the worm making H2O2? It has been engineered to do so. No, not by genetic engineering, but by materials science. The scientists designed a protective sheath for the worm. Then, they bound the enzyme glucose oxidase (GOX) to the surface. GOX is a well-known enzyme; it oxidizes glucose to gluconic acid -- and makes H2O2 as a by-product.
The big idea is that the worm would seek out cancer cells, and then oxidize readily available glucose to make H2O2, which would kill the cancer cells.
Much of the work in this article is about engineering the worms -- developing the protective sheath and a method for binding the enzyme. The worms survive all this quite well.
The approach is generalizable. The worms could be engineered to deliver other cargo for other applications.
It is one of the first studies of surface engineering of organisms other than microbes. It's all quite intriguing -- and quite preliminary at this point.
What does NAGOX mean? They don't say. The GOX part obviously refers to glucose oxidase. It is possible that the N and A refer to nematode and Anisakis.
News stories:
* These newly-engineered parasitic worms can kill cancer cells -- The worms are found in nature and are attracted to cancer cells. (Loukia Papadopoulos, Interesting Engineering, July 10, 2022.) (Incorrectly names the journal, but the link is fine.)
* Scientists Have Created Worms That Can Kill Cancer Cells. (SciTechDaily (Osaka University), July 7, 2022.)
The article, which is open access: Nematode surface functionalization with hydrogel sheaths tailored in situ. (Wildan Mubarok et al, Materials Today Bio 15:100328, June 2022.)
Another case of using glucose oxidase to make H2O2 for killing: Why are the bees dying? (January 26, 2010).
More glucose oxidase: A smart insulin patch that rapidly responds to glucose level (October 26, 2015).
Another potentially therapeutic hydrogel: What if infection triggered a treatment? (August 2, 2022).
Among many posts about nematodes: Worm count (August 27, 2019). Links to more.
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Cancer. It includes an extensive list of relevant Musings posts.
July 20, 2022
Briefly noted... X-ray analysis of a remarkably preserved fossil cephalopod, related to the vampire squid
July 20, 2022
Cephalopods (octopus and such) lack a hard skeleton; well-preserved fossils are rare. A new article reports X-ray analysis of 164-million-year-old specimens of Vampyronassa rhodanica, which is thought to be a close relative of the modern vampire squid (see background post noted below). Simply doing it is noteworthy. And the work suggests that this species was an active feeder.
* News stories:
- Vampire Squid's Ancestor was Active Hunter, New Study Suggests. (Sci-News.com, June 23, 2022.)
- X-rays reveal the strange appendage this ancient vampire squid used to hunt. (Grant Currin, Interesting Engineering, June 24, 2022.) Story title is misleading, but this is a good item, and includes a good interview with the lead author.
* The article, which is open access: Exceptional soft.tissue preservation of Jurassic Vampyronassa rhodanica provides new insights on the evolution and palaeoecology of vampyroteuthids. (Alison J Rowe et al, Scientific Reports 12:8292, June 23, 2022.)
* A post about the vampire squid: Quiz: What is it? (November 20, 2012).
* More about cephalopods: Sleep stages in octopuses -- do they dream? (July 13, 2021). Includes an extensive list of posts about octopuses and other cephalopods.
Would moth wings make good sound absorbers?
July 19, 2022
Does this look like it might be good sound-absorbing material?

|
That's a scale from a moth wing. (Scanning electron microscopy.)
The scale bar (lower left) is labeled 2 micrometers.
(The labels on the wing are to identify two measurements: IR = inter-ridge, CR = cross-rib.)
The moth is Antheraea pernyi.
This is Figure 2c from a new article.
|
The next figure shows some data on the matter...
The idea is to measure the reflection of sound from a piece of metal. In some cases, a single moth wing -- or part of one -- was attached to the metal. The graph shows the amount reflected (y-axis) vs frequency (x-axis). The lower the value, the better (more sound absorption).
The top curve (black) is for the bare metal ("disc only", referring to the metal disc).
The lowest curve (blue; best sound absorption) is for the metal disc with an intact moth wing attached.
The other two curves have some wing layers; see the key. They give intermediate results.
This is Figure 3a from the article. (I partially labeled the x-axis, which was cut-off.)
|
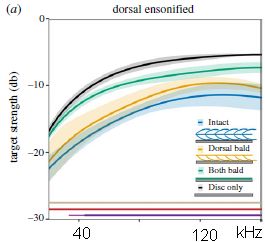
|
The wings are quite good sound absorbers, especially considering how thin and lightweight they are. The wing layers (main membrane and scales) contribute; the top figure only hints at the sound absorption by the wings. The work here extends what was known about how the wings absorb sound in free space to how they work with sounds reflected off a hard surface.
Why might moth wings do this? As protection against bats, which find prey by echolocation. (That also explains the focus on high frequencies in the current article.)
The authors have developed a computer model of how the moth wings absorb sound. The model considers the various structural elements, as well as air gaps.
They suggest that the design of moth wings might offer clues to inspire the rational development of better sound absorbing materials. Moth-inspired wallpaper could be in your future.
The final sentences of the article (in Conclusions): The mechanism of sound absorption is unclear but is likely a combination of the mechanical absorption of the scales coupled with some dissipation through thermal and viscous effects brought about by the interaction of the scales, wing membrane and air movement through the scales. It is hoped that this understanding of the absorption mechanisms of scales of the moth wing will inspire the next generation of acoustic metamaterial sound absorbers.
News story: Moth wing-inspired sound absorbing wallpaper in sight after breakthrough. (Phys.org (University of Bristol), June 14, 2022.)
The article, which is open access: Moth wings as sound absorber metasurface. (Thomas R Neil et al, Proceedings of the Royal Society A 478:20220046, June 2022.)
More about moths and bats: Warfare: the tymbal (September 3, 2009).
For more about bio-inspiration, see my Biotechnology in the News (BITN) topic Bio-inspiration (biomimetics). It includes a listing of Musings posts in the area.
Virus infection may make you more attractive to mosquitoes
July 16, 2022
A new article provides evidence that some viruses may promote their own transmission by making their host more attractive to the mosquitoes that transmit them.
The article is quite complex. We'll look at the evidence for a couple of steps, and then outline the rest of the story.
The work was done with Zika and dengue viruses, two members of the flavivrrus group. Such viruses are transmitted by mosquitoes.
Step 1. Mosquitoes are attracted to virus-infected hosts.
The following figure shows some of the evidence...
The test is logically simple, though the scientists developed a fairly complex apparatus to allow carefully controlled experiments. The big idea is that mosquitoes were offered two hosts (mice). One was infected with Zika (right side; brown). The other host was "healthy" (not virus-infected; left; black). The x-axis is labeled "choice".
The top row is for day 0 (the day of infection). The bars are of equal length. That is, the mosquitoes chose infected and uninfected hosts equally. That is also true at 2 dpi (days post infection).
However, as the infection continued, the mosquitoes increasingly chose the infected mice (last two rows, 4 and 6 dpi).
This is Figure 1B from the article.
Similar results were obtained using mice infected with dengue virus; Figure 1C.
|
Step 2. Identifying the attractant.
One might guess that the mosquitoes are being attracted by something in the air -- something volatile made by the mice after infection.
To test this, air from around the mice was tested. A number of chemicals seemed to have different levels depending on whether or not the mice were infected. These chemicals were tested, in part by a lab test of whether mosquito antennae responded to them. That work pointed to one chemical, acetophenone, as the main suspect.
With a suspect chemical in hand -- almost literally -- it could be directly tested. The following figure shows one such test -- on human hands.
The logic of the test here is similar to the one above. But in this case, the mosquitoes were offered human hands, with or without the chemical being tested. The amount of acetophenone is shown at the left.
The results are clear... the more acetophenone on the hand, the more the hand was chosen by the mosquitoes.
This is part of Figure 2E from the article.
Similar results were obtained with the acetophenone on the backs of mice; Figure 2D.
|
So far, we have shown evidence that mosquitoes are attracted to hosts infected with a flavivrrus, and that the chemical acetophenone is the attractant molecule.
The structure of that chemical is shown at the right.
This is from the graphical abstract of the article.
|

|
There is more... How is the attractant made? How does the virus affect attractant production? The scientists go on to show that it is made by certain bacteria on the skin. Further, the virus infection affects the types of bacteria present, and they have some evidence on the signaling molecules involved. That is, virus infection causes changes in the skin microbiome, leading to an increase in bacteria making acetophenone.
The work on how the microbiome change occurs leads to the suggestion that a vitamin A derivative might be helpful in reducing transmission of these viruses. Their preliminary results are encouraging, but there are potential pitfalls with using vitamin A. Another possible follow-up might be to try blocking the response of mosquitoes to acetophenone. Also, detection of acetophenone might be a useful diagnostic for these flavivrrus infections.
But that is enough detail for one post.
News stories:
* Infection With Tropical Viruses Can Make You Smell Tastier to Mosquitoes. (Sarah Whelan, Technology Networks, June 30, 2022.)
* Some viruses make you smell tastier to mosquitoes -- Dengue and Zika viruses alter the microbiome in both mice and humans to attract mosquitoes and spread to new hosts. (Science Daily (University of Connecticut), June 30, 2022.)
* Viruses can change your scent to make you more attractive to mosquitoes, new research in mice finds. (Penghua Wang, The Conversation, June 30, 2022.) From one of the authors of the article.
The article: A volatile from the skin microbiota of flavivirus-infected hosts promotes mosquito attractiveness. (Hong Zhang et al, Cell 185:2510, July 7, 2022.) Check Google Scholar for a freely available copy.
Among posts on attracting mosquitoes...
* A trap to attract -- and kill -- mosquitoes (October 26, 2021).
* Malaria-infected mosquitoes have greater attraction for people (May 28, 2013). Some articles on this topic are cited by the current article.
More about the viruses is on my page Biotechnology in the News (BITN) -- Other topics under Dengue virus (and miscellaneous flaviviruses) and Zika. Each of those sections includes a list of Musings posts.
July 13, 2022
Briefly noted... How intelligent is your planet?
July 13, 2022
From the abstract of a recent article... "We argue that explorations of planetary intelligence, defined as the acquisition and application of collective knowledge operating at a planetary scale and integrated into the function of coupled planetary systems, can prove a useful framework for understanding possible paths of the long-term evolution of inhabited planets including future trajectories for life on Earth and predicting features of intelligentially steered planetary evolution on other worlds." It is best to take such things as provocative models, which one explores to see what happens. Proceed at your own risk.
* News stories:
- Can Planets Be Intelligent? (Sci-News.com, February 22, 2022.)
- Can a planet have a mind of its own? Thought experiment. (Science Daily (University of Rochester), February 16, 2022.)
* The article, which is open access: Intelligence as a planetary scale process. (Adam Frank et al, International Journal of Astrobiology 21:47, April 2022.)
* The approach here can be seen as related to the Gaia model. I have noted this item on the Musings supplemental page Gaia and James Lovelock. By the way, Lovelock turns 103 later this month (July 26).
* Also see... Briefly noted... James Lovelock, 1919-2022 (August 1, 2022).
Is being tall bad for your health?
July 12, 2022
There have been hints of such effects before... tall people may be more subject to certain conditions. (For some conditions, tall people may be at less risk.)
A new article reports an extensive statistical analysis of about 280,000 people in a database (of military veterans, in the US). The idea is to look for a correlation between genetic height (height as predicted using known genetic factors) and specified conditions.
They also do analyses using actual height. An advantage of genetic height is that it eliminates environmental factors that lead to actual height not being the genetic height.
Here are the basic findings...

The records were divided by racial/ethnic group, based on self-identification. Only two such groups were large enough to give statistically significant results: non-Hispanic White and non-Hispanic Black. These are designated EA and AA, presumably for European Americans and African Americans, respectively.
The scoring is the odds ratio, OR. It is the probability of having the trait relative to average. (It is for a one standard deviation change in height. I think; not entirely clear.) The x-axis is the OR for the AA (African Americans) in the database. The y-axis is the same for the EA (European Americans)
For about a hundred traits, there seemed to be a significant association of the condition with genetic height for the EA population (the larger sub-set). All of those are shown on the graph above, plotting the OR for EA vs the OR for AA,
You can see that many of the points are near the diagonal line, meaning that they showed similar OR for the two groups. This suggests a consistent effect across two racial groups.
The points are color-coded by their statistical significance in the AA data. (All the points were significant for EA.) The green and blue points showed significance for AA; the orange points did not. At least roughly, the color coding by significance agrees with the general visual impression of simply looking for points near the diagonal line.
About 90% of the records were for males.
This is part of Figure 1B from the article. (The A and B parts of the figure are not labeled, though they are referred to in the figure legend and main text.)
|
It is best to think of such studies as generating leads, which can be followed up. But the big picture is that there seem to be numerous conditions for which a tall person is at greater risk (and some where they are at lower risk). The article (and news stories) discuss some of the specifics, but we'll skip that here. The authors are properly cautious in their own conclusions.
News stories:
* Study: Height May Be Unrecognized Risk Factor for Several Common Conditions in Adults. (Sci-News.com, June 6, 2022.)
* How Your Height May Influence Your Risk of Disease. (Sarah Whelan, Technology Networks, June 16, 2022.)
* Height may be risk factor for multiple health conditions. (Science Daily (Veterans Affairs Research Communications), June 2, 2022.)
The article, which is open access: A multi-population phenome-wide association study of genetically-predicted height in the Million Veteran Program. (Sridharan Raghavan et al, PLoS Genetics 18:e1010193, June 2, 2022.)
Posts about human height include...
* The rising rate of caesarean section births: an intriguing correlation (April 15, 2019).
* The modern pygmies of Flores Island (November 6, 2018).
* (Most references to height in Musings seem to be about bar heights.)
Inhibiting the TMPRSS in your nose to protect against respiratory viruses (COVID, flu)
July 11, 2022
It is actually rather logical, and a recent article suggests it might work. The current experimental work is with the SARS-2 virus (COVID-19) in mice, but there is reason to think it may be of wider use. (The work began, prior to COVID, with influenza.)
The key idea is that infection with this virus (and some others) is more complex than we usually say. You know about the SPIKE protein of the SARS-2 virus binding to a receptor on your cells, to gain entry. But there is one step before that: the SPIKE protein needs to be cleaved. TMPRSS2 is a protease, an enzyme on the surface of your cells that cleaves SPIKE. So, it makes sense... inhibit TMPRSS2, and that should prevent infection.
The first phase of the work was to develop an inhibitor of the enzyme. We will focus here on one inhibitor that the scientists developed, the one they consider the best so far; it is called N-0385.
The next question is whether the protease inhibitor actually affects the virus. Here are the results of one test,,,

|
The inhibitor was tested against five strains of SARS-2 virus: an ancestral strain, plus four variants.
The graph shows a measure of the infection (y-axis) vs drug dose (x-axis, log scale).
As a generality, the curves reach 50% effectiveness at about -8 on the x-axis scale. That means 10-8 M, or 10 nM. The table shows the actual values for EC50, the drug concentration needed for 50% inhibition.
|
In this test, virus infection was monitored by measuring the amount of viral RNA. It is shown as a percentage of the control (y-axis).
This test is with a line of human lung epithelial cells in lab culture.
This is Figure 3d from the article.
|
That test shows that the drug is quite potent, and that it acts against a range of variants of the original virus.
The activity does vary, over about a 10-fold range, between the strains. We must wonder how other variants would fare, or whether the virus can develop substantial resistance to the drug.
The drug does not act directly on the virus, but rather on a protein from the host. Nevertheless, the virus might mutate so that it no longer needs the enzyme that is being inhibited. For example, it might infect without being cleaved, it might cleave spontaneously, or it might be cleaved by another enzyme.
The test shown above measures the effect on the virus. The next test measures the effect on the host...
Start by comparing the two solid curves. The black curve shows the survival of infected mice over time after a mock treatment ("saline"). The red solid line shows the survival with the drug treatment. You can see that the drug greatly improved survival.
The dashed curve is for a modified drug, which the scientists expected would be inactive. It was.
|

|
The drug treatment here was by a nasal spray on days -1 to +6 (day 0 is the day of virus infection).
The inactive drug has (OH) in the name. The active drug has a carbonyl (ketone) group, which they thought was important. It was reduced to -OH to make the inactive analog.
The test was done with humanized mice. The mice carried the human version of the ACE2 gene; that protein serves as the viral receptor.
This is Figure 4f from the article.
|
There is much more in the article, of course. The work suggests that N-0385 usefully treats COVID infections, and is active against all COVID strains tested so far. It should be given early, and may have benefit prophylactically. The drug is easy to administer.
Trials in humans are planned. It is possible that the drug could become available later this year, though that seems rather optimistic.
As noted at the top, the work on TMPRSS2 inhibitors started with influenza as the target. The drug may be useful against other viruses, too.
News stories:
* Promising nose spray could prevent and treat COVID-19. (Krishna Ramanujan, Cornell Chronicle, March 28, 2022.)
* TMPRSS2 inhibitor found to be a novel and effective therapeutic option against pan-variant SARS-CoV-2 infections. (Pooja Toshniwal Paharia, News-Medical.net, April 1 2022.)
The article, which is open access: A TMPRSS2 inhibitor acts as a pan-SARS-CoV-2 prophylactic and therapeutic. (Tirosh Shapira et al, Nature 605:340, May 12, 2022.)
What does TMPRSS2 mean? Transmembrane protein serine protease 2. If that doesn't seem quite right, so be it.
Among posts about proteases and agents that affect them...
* Inter-plant communication via the Cuscuta parasite (September 15, 2017).
* Killing persisters -- a new type of antibiotic (January 3, 2014).
Among posts about anti-viral agents...
* An antiviral coating for medical textiles (July 12, 2020).
* Could we treat COVID by driving it to an error catastrophe? (June 30, 2020).
There is a BITN section for SARS, MERS (coronaviruses). It includes a list of Musings posts in the field.
Posts on flu are listed on the supplementary page Musings: Influenza.
Growing food with artificial photosynthesis?
July 9, 2022
Ordinary photosynthesis uses solar energy to "fix" CO2 from the air into bio-molecules. Perhaps we could separate those steps.
There is some CO2 fixation without photosynthesis in normal biology. It is usually considered to be a small fraction of overall CO2 fixation. A post about this is at the end.
The following figure presents the idea, as developed in a new article...
Part a (left) shows the first step in the proposed process: fix CO2. It is a chemical process, not biological. It requires energy, but that can be our choice of energy. The process shown here uses solar energy to drive an electrolysis reaction. The electrolysis converts the CO2 into acetate, which is a good food for many organisms.
Part b (center) shows growing a variety of organisms on that acetate from part a. These are all ordinary organisms, which can be used as or in food products (part c; right).
The energy source for part a is open. They show it here as solar-based electricity, but any electricity will do.
This is Figure 1 from the article.
|
Why would we want to do this? Well, it is more flexible. Using CO2 has been separated from sunshine.
More specifically, the scientists argue that their process is more efficient-- uses solar energy more efficiently. That is outlined in the following figure...
The figure shows what happens to the solar energy in two processes: ordinary biological photosynthesis (left), and their artificial photosynthesis (right).
Look at the bottom, where they show how much biomass is made. Four times as much, using their artificial photosynthesis process. That is their point.
That result of a four-fold improvement is for algae. A similar analysis for yeast, which is not a primary producer, suggested an 18-fold increase in efficiency.
This is Figure 5 from the article.
|
That's the big picture. They propose fixing CO2 by electrolysis, to make acetate, which serves as the basis of food production. This process is more efficient (and more flexible) than the traditional process using biological photosynthesis.
Much of the experimental work went to designing an electrolyzer system that led to a product suitable to be fed to plants. Acetate is a two-carbon chemical that fits right into ordinary metabolism; further, it is soluble and non-toxic. (They found some toxicity with higher plants.)
There is much detail in the article, including data to support their claims. They are far from any practical implementation, but it certainly is an interesting approach. It has the potential to increase food production without increasing the land needed for agriculture. It might also be of interest for space travel.
News stories:
* A new breakthrough in biology allows scientists to grow food without sunlight -- Artificial photosynthesis could be 'a paradigm shift for how we feed people.' (Chris Young, Interesting Engineering, June 24, 2022.)
* Producing food without sunlight through artificial photosynthesis. (News-Medical.net (University of Delaware), June 29, 2022.)
* Artificial photosynthesis can produce food without sunshine. (Nanowerk News (University of California - Riverside), June 23, 2022.)
* News story accompanying the article: Food synthesis: Artificial photosynthesis of food from CO2 -- Food production in a given solar footprint is limited by the efficiency of natural photosynthesis. Now, a hybrid electrochemical-biological artificial photosynthesis system demonstrates the potential for food synthesis from CO2 and electricity, enabling a paradigm shift in food production. (Tuo Wang & Jinlong Gong, Nature Food 3:409, June 2022.)
* The article, which is open access: A hybrid inorganic-biological artificial photosynthesis system for energy-efficient food production. (Elizabeth C Hann et al, Nature Food 3:461, June 2022.)
A post noting the natural role of CO2 fixation without photosynthesis: Is photosynthesis the ultimate source of primary production in the food chain? (April 2, 2017).
Another system of artificial photosynthesis: More from the artificial forest with artificial trees (August 31, 2015). There are multiple posts on this system.
There is more about energy on my page Internet Resources for Organic and Biochemistry under Energy resources. It includes a list of some related Musings posts.
July 6, 2022
Briefly noted... What color are sunflowers, and why?
July 6, 2022
If you think that they are yellow, or that all sunflowers are about the same... It is well known that they vary in color, in the ultraviolet; pollinators can detect that. An article from earlier this year does genetic analysis of how the UV pigment is controlled, and also suggests that the pigment plays an important role in water retention. The results could have implications for developing plants that are more drought tolerant.
* News story: Sunflowers' invisible colors help them attract bees and adapt to drought. (Science Daily (University of British Columbia), January 18, 2022.) Links to the article, which is open access.
* Direct link to the article, which is open access: Genetic basis and dual adaptive role of floral pigmentation in sunflowers. (Marco Todesco et al, eLife 11:e72072, January 18, 2022.)
* More about sunflowers: Why growing sunflowers face the east each morning (November 8, 2016).
ROOOH
July 5, 2022
Three oxygen atoms together, -O-O-O-. Not common. Probably the only familiar chemical with that feature is ozone, and it is highly reactive. But there is interest in the possibility of such organic hydrotrioxides, R-O-O-O-H, being made in the atmosphere and being potent oxidizers there. (As usual, R can be "anything"; the requirement is that it has a C atom that connects to the O system.)
A recent article provides evidence for such compounds in a lab-scale model of atmospheric chemistry.
Here is one hydrotrioxide of interest...

|
Compound III (at right, in red) is a hydrotrioxide. At the top of the N is attached -CH2-OOH. That is a hydroperoxymethyl group. There is another of those at the bottom of the N. But the group to the right has one more O: -CH2-OOOH, with hydrotrioxy on the methyl group.
|
How is it made? From compound I (at the left). I differs from III in having -CH2-OO·. Note the raised dot, indicating it is a radical, with an unpaired electron. Compound I reacts with ·OH, the hydroxyl radical. Joining those two radicals gives compound III.
Where does I come from? It doesn't look common. In the model system here, the scientists add trimethylamine, N(CH3)3. It gets oxidized under the experimental conditions, to I (among other things).
Hydroperoxy- could be called hydrodioxy-.
This is part of Figure 1 from the article. The full figure shows some competing reactions.
|
The following figure shows evidence that compound III, the hydrotrioxide, is made in their system. Caution, the figure is complex; we'll focus on the evidence for the main point, the production of III.

The figure shows the amounts of various chemicals present in the system, based on mass spectrometry analysis of the artificial atmosphere.
Both y-axis scales show amounts of chemicals present; both are log-scales. The main scale, on the left, simply shows relative amounts.
The x-axis scale shows the amount of one key component of the system, IPN (isopropyl nitrite). The role of this chemical is to generate the ·OH, the hydroxyl radical that is one key reactant for making III. This scale is also log-scale, and is in molecules/cm3.
The red in the figure is for the compound of interest, III. The graph shows that the amount of III increases steadily as the amount of ·OH-generating IPN is increased.
If you want more detail... The reactant I is also shown; it is the top line. The amount of I is also increasing, showing a role for the oxidizing reactant (x-axis) in making I. The slope of the line for III is greater than the slope for I; this shows that the ratio of III to I is increasing. At the highest concentration of IPN used, this ratio is about 1%.
As noted, the role of the IPN is to generate ·OH. The amount of ·OH made is shown by the blue curve; read it on the right-hand y-axis, which is in molecules/cm3. This curve seems to be based on a theoretical calculation, not a measurement.
This is Figure 2A from the article.
|
They also show the production of hydrotrioxides from important atmospheric pollutants, such as isoprene and dimethyl sulfide.
The work shows that ROOOH compounds can be made in a model system designed to be like the atmosphere. That doesn't prove they are made in the real atmosphere, but it is a step. It also gives a base for further work on their properties under atmospheric conditions.
The scientists estimate that these compounds have lifetimes on the order of minutes to hours in the atmosphere.
Organic hydrotrioxides are known to organic chemists; they can be made under well-defined conditions, and are useful in the lab. What is new here is implicating them in atmospheric chemistry. More specifically, what is new is providing experimental evidence on the matter. There is some confusion on this point in some of the news converge.
News stories:
* Researchers detect a chemical they thought was too unstable to survive -- And it might have a hidden effect on climate change and human health. (Ameya Paleja, Interesting Engineering, June 3, 2022.)
* New Class of Unusual Reactive Substances Detected in Atmospheric Chemistry. (SciTechDaily (Leibniz Institute for Tropospheric Research (TROPOS)), June 5, 2022.) Links to another story based on the press release from the University of Copenhagen.
The article: Hydrotrioxide (ROOOH) formation in the atmosphere. (Torsten Berndt et al, Science 376:979, May 27, 2022.)
A recent post about atmospheric chemistry, also involving radicals: Briefly noted... how dust reduces ozone in the atmosphere (June 29, 2022).
Among posts on other pollutants mentioned...
* The importance of HPMTF in the atmosphere (June 9, 2020). Dimethyl sulfide.
* Interaction of pollution sources: Can the whole be less than the sum of the parts? (March 9, 2019). Isoprene.
Making hydrogen visible
July 2, 2022
Hydrogen gas is invisible. Odorless, too. And explosive. As we make moves toward a hydrogen economy, how will we know about hydrogen leaks or such -- before that last step?
A new article reports a way to make hydrogen visible to the naked eye.
Here is what the test looks like...
Part a (upper left) shows the set-up. Simple. A vial containing a powder. A gas stream is directed into the vial, and ...
The other parts show examples of what happens as H2 gas is or is not added. In each case, there is a labeled timeline across the top, and some photos. Note that the photos are labeled by time, and do not always line up well with the timeline above.
In part b (upper right), you can see that the material has three colors: purple, pink, and colorless. It starts purple. When H2 is added, it turns pink then colorless, over about 2 seconds. When H2 is removed, it returns to pink (but not purple).
Part c shows the results from repeated H2 on/off cycles. In each case, the color changes within a few seconds, indicating that H2 is or is not present.
This is Figure 3 from the article.
|
Those results show that the indicator undergoes a reversible color change, easily visible to the naked eye, within a few seconds, depending on whether or not H2 is present.
There is a movie file posted with the article: Movie file (one minute; no sound). It shows the test from which the images in part c, above, were taken.
The sensitivity of the indicator is not discussed in the article. One test is done with a gas feed that is about 17% H2 (by volume). Presumably, lower concentrations would give at least noticeable color changes, but there is no information about the limit.
How does the indicator work? It is based on the reversible reduction of a dye molecule in the presence of H2. The following figure summarizes the chemistry involved...
Start with the two chemicals to the right, #2 and #3. The reaction arrows show that these interconvert depending on whether or not H2 is present. #2 is pink, and #3 is colorless. That is the basis of the indicator.
The coloring in the figure makes it hard to see some detail, but the 2 H atoms of H2 end up on the N at the top and the O at the lower right. The H on the N breaks the conjugation (alternation of single and double bonds) across the entire molecule; that causes loss of color.
Chemical #1, at the left? Resazurin. That is actually what they add to the material. It requires a preliminary reaction to convert it to the active indicator, #2. #1 itself is purple; recall that first purple-to-pink change in the first figure.
This is Figure 5a from the article. I added numbers by the names of the three chemicals.
|
Much of the article is about practical aspects of the implementation, including the role of the catalytic sites, support materials, and water in the particles.
It's promising... a simple visual indicator for the presence of hydrogen gas. Making it required consideration of the chemistry, as well as putting it together in a practical device.
News story: Researchers develop a sensor to make hydrogen visible. (Nanowerk News (Friedrich-Alexander-Universität Erlangen-Nürnberg), March 21, 2022.)
The article, which is open access: Supraparticles for Bare-Eye H2 Indication and Monitoring: Design, Working Principle, and Molecular Mobility. (Jakob Reichstein et al, Advanced Functional Materials 32:2112379, May 25, 2022.) The Introduction discusses alternative H2 detection systems.
Among recent posts about hydrogen...
* Separating isotopes of hydrogen using a MOF (April 18, 2022).
* Was hydrogen the energy source for the origin of life? (February 1, 2022).
... and about sensors... A new sensor for barium ions -- could it lead to a better understanding of neutrinos? (September 20, 2020).
June 29, 2022
Briefly noted... how dust reduces ozone in the atmosphere
June 29, 2022
It has long been noted that regions of the upper atmosphere with a lot of dust tend to have low ozone. Why? A recent article provides evidence for a complex but important story. Here is a brief version of what the scientists think happens... Iodine, possibly in the form of iodate, can be transported on dust. In the atmosphere, it can form the iodine monoxide radical, IO·. (Note the dot in the formula, to help show this is a radical; there is no charge on it, but there is an unpaired electron.) IO· can react with ozone, O3, and it is actually quite good at it. Thus dust-borne iodine can cause serious damage to the ozone layer of the upper atmosphere.
* News story: Ozone depletion due to release of iodine dust -- Iodine in Desert Dust Destroys Ozone. (Amit Malewar, Tech Explorist, December 24, 2021.) Links to the article, which is open access.
* Direct link to the article, which is open access: Ozone depletion due to dust release of iodine in the free troposphere. (Theodore K Koenig et al, Science Advances 7:eabj6544, December 22, 2021.)
* Also see:
- ROOOH (July 5, 2022). More atmospheric radicals.
- Geoengineering: the advantage of putting limestone in the atmosphere (January 20, 2017). This post is about the effects of atmospheric particulates on ozone depletion. The authors of the current article caution that they have found an additional complexity in the story.
What do we learn from the sulfur isotopes in the California vineyards?
June 28, 2022
A new article reports an analysis of the sulfur in Napa County, in the heart of Northern California's wine country.
Here's a summary of what the scientists found...

Two measurements were made for each sample. The concentration of sulfate is shown on the x-axis. The isotope analysis of the sulfur in that sulfate is shown on the y-axis. (As common with such isotope analyses, it is shown as a difference from a reference sample. That detail doesn't matter much. What matters is that the y-axis reflects the amount of S-34 in the sample.)
Each symbol is coded two ways, as shown in the double-key at the right. The shape of the symbol indicates the type of sample (e.g., soil or stream). We can skip that, at least to start.
The color of the symbol shows the type of area the sample is from. Of particular interest: purple for samples from the vineyards, and green for lands with natural vegetation, such as forests.
Observations:
- The sulfate levels in the vineyard samples (purple) vary widely; many are high. In fact, all the points above about 32 on the x-axis are from vineyards. In contrast, the samples from lands with natural vegetation (green) have low values, almost all below about 12.
- The pattern is similar for the S-34 (y-axis). Most (but not all) of the samples from the vineyards are above 10 on this scale. All of the samples from natural vegetation areas are below 10.
This is Figure 2 from the article.
|
The big picture is that the vineyard samples are high in sulfate. Why? and does it matter?
The why is simple. It is due to the use of sulfur as a fungicide on the grapevines. Sulfur? Yes, the yellow stuff, pure elemental sulfur, commonly called agricultural sulfur. It has long been used for this purpose. It is now the top pesticide used in California agriculture. That S gives a distinct isotopic signature, which is apparent in the analyses above.
Sulfur does protect the crop, but much of it ends up being washed off into the soil, ultimately in the oxidized form of sulfate ion. It also ends up in the Napa River (black symbols), which promotes its distribution.
The current work shows that the contribution of the agricultural S to the surrounding environment is significant. How much it matters is open at this point, but there is an increasing sense that we should at least be aware of such issues, and perhaps should mitigate them. For example, we might try to reduce the amount of S added to the crop; there is little basis for the current procedure.
News story: Toward more sustainable wine: Scientists can now track sulfur from grapes to streams. (Phys.org (University of Colorado at Boulder), May 24, 2022.)
The article, which is open access: Sulfur isotopes reveal agricultural changes to the modern sulfur cycle. (Anna L Hermes et al, Environmental Research Letters 17:054032, May 2022.)
Some wines are labeled "no sulfur added". That has nothing to do with the current issue. It refers to whether or not sulfite, SO32- (or, equivalently, sulfur dioxide), is added to the wine, as a preservative. (Sulfate, which is the major product of the sulfur used on the vines as a fungicide, is SO42-.)
Previous post about grapevines:
* A half-millennium record of climate change, from the grapes of Burgundy (November 9, 2019).
* Acne, grapevines, and Frank Zappa (August 1, 2014).
A post based on data from GRAPES: High-voltage thunderstorms: how high? (April 29, 2019).
More about fungicides:
* Keanu Reeves and a broad-spectrum fungicide (March 6, 2023).
* Largest field trials yet... Neonicotinoid pesticides may harm bees -- except in Germany; role of fungicide (August 20, 2017).
More about pesticides: The role of ants in agricultural pest control (September 12, 2022).
Posts on broader agricultural issues include... Evaluating the sustainability of food systems (December 5, 2022).
My page of Introductory Chemistry Internet resources includes a section on Nuclei; Isotopes; Atomic weights. It includes a list of Musings posts, some on measurements of stable isotopes.
A bio-marker for SIDS?
June 27, 2022
SIDS = sudden infant death syndrome. A baby, thought to be healthy, just dies (typically, while asleep).
A new article reports a possible bio-marker for SIDS. If it is validated, it would at least focus attention on certain babies that are at higher risk for SIDS.
The work involves an enzyme called butyrylcholinesterase. It can be tested in dried blood spots, which are routinely obtained from newborns. That's the basis of what was done here. The authors tested samples from the hospital archive -- samples from babies who died of SIDS, babies who died from other causes, and healthy controls.
The following figure summarizes the key results...
For each baby, the enzyme activity in the dried blood sample was measured. That result is shown here as the difference for each baby from the average level.
The data set is for 26 babies who died of SIDS, and 30 babies who died of other causes.
There is one bar for each baby tested; the bars are in order by the enzyme level. Further, the bars are color-coded: red for SIDS cases, blue for non-SIDS.
There is a pattern: no babies with an enzyme level above about 2 on this scale died of SIDS.
This is Figure 4 from the article.
|
That result suggests that a low level of the enzyme means that a baby is at a higher risk of SIDS.
Importantly, the work does not say anything about this enzyme having any role in the disease. There is nothing in the study about that. A bio-marker is something measurable that correlates with the condition; that is the claim here.
Since the enzyme is part of the nervous system, it is tempting to wonder about its role in the disease. The enzyme is involved in arousal; babies with lower levels of the enzyme might be deficient in waking from a respiratory distress. But that is for further work to explore. This article provides no evidence on the role of the enzyme.
Is this a useful bio-marker? It might be; it is certainly easily measured. The first question is whether the result can be verified, preferably in more complete studies. The quantitative results above suggest that its predictive power is modest, but it is a start.
Bottom line... It is an interesting result, which needs to be followed up. That is what preliminary results are about.
The enzyme butyrylcholinesterase, despite its name, hydrolyzes acetylcholine. Its more famous cousin is acetylcholinesterase; that enzyme could not be measured from the available blood samples.
News stories:
* First Potential Biomarker for Sudden Infant Death Syndrome Identified. (Ruairi J Mackenzie, Diagnostics from Technology Networks, May 17, 2022.)
* Butyrylcholiesterase may be key biomarker in SIDS identification, prevention. (Katie Hobbins, Contemporary Pediatrics, May 17, 2022.)
* Expert reaction to study attempting to identify cause of and biomarker for Sudden Infant Death Syndrome (SIDS). (Science Media Centre, May 13, 2022.) Both comments stress the need for caution in interpreting the finding.
The article, which is open access: Butyrylcholinesterase is a potential biomarker for Sudden Infant Death Syndrome. (Carmel Therese Harrington et al, eBioMedicine 80:104041, June 1, 2022.)
A previous post on SIDS: Sudden infant death: a genetic factor affecting breathing? (April 30, 2018).
Posts that mention the neurotransmitter acetylcholine:
* The importance of choline as a dietary nutrient (April 15, 2023).
* An artificial neuron? (November 6, 2015).
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Brain. It includes a list of brain-related posts, including some that are more generally about the nervous system.
Selenium and neurodegenerative diseases?
June 25, 2022
Sodium selenate, Na2SeO4. A few years ago, scientists found that it activates an enzyme that removes the phosphate group from phosphorylated tau protein.
Tau is a brain protein that has bean implicated in various neurodegenerative diseases. The phosphorylated form of tau is thought to be particularly important.
Is it possible that selenate could slow the progression of a neurodegenerative disease by dephosphorylating tau?
That's the question behind a recent article testing the effect of selenate on people with behavioral variant frontotemporal dementia -- bvFTD, for short. It's a very small study, but the results are at least intriguing.
It's a phase I trial, where the main goal is to test the safety of the proposed treatment. That's simple: the drug was well tolerated (as it had been in some other tests). The test involved 12 people treated for a year, and there were no serious adverse events attributable to the drug. (There were various "minor" effects, some of which are listed in the article (Table 2). These are effects that would be noticed, but would not preclude a treatment if it had meaningful benefit.)
A phase 1 trial is not primarily about benefit, but does include some observations. The following figure offers a glimpse of the effect of the drug on the course of the disease.

|
The graph shows changes in two measures of the disease over the treatment time. The x-axis is for percent brain volume change (PBVC; measured with MRI). The y-axis is for a score on a test of mental function (NUCOG, a test of COGnition). For both measures, patients with bvFTD typically show negative changes; zero is good.
Each point is for one patient, showing how they progressed by these two measures.
|
Observations...
- The points seem to form two clusters.
- One cluster, to the lower left, shows negative values for both measures. This is about as expected for patients with normal disease progression.
- One cluster, to the upper right, shows higher values -- closer to zero, though mostly still negative -- for both measures. The disease progressed less than typical in these people.
The two colored points are for two patients with specific known mutations. Since there was only one of each type in the study, no generalizations are possible.
This is Figure 2A from the article.
|
Are those two clusters responders and non-responders? Over half of those treated seemed to benefit from the sodium selenate (showing less loss of both brain volume and function).
The test is too limited to reach any big conclusions; there are only 12 patients, and no controls. But it is intriguing; more testing is clearly warranted -- and is in progress.
The article contains another intriguing result. Is it possible to predict which patients will respond to the treatment? Here is what one analysis showed...
The graph shows the likelihood of response (y-axis) plotted against a measure of tau on the x-axis. More specifically, what is plotted here is the ratio. at baseline (the beginning of the trial). of phosphorylated tau to total tau in the cerebrospinal fluid (CSF).
The curve suggests there is a trend: that people with a higher percentage of phosphorylated tau are more likely to respond to the treatment.
That's a lot of fancy graph for a very small amount of data. The pattern must be taken as only a hint, but it is interesting.
This is Figure 3C from the article.
|

|
It's all very preliminary. Remember, the main point of a phase 1 trial is safety. The treatment seems to have passed the safety test rather well, and it also offers hints of some benefit. There is some logic to the story... Phosphorylated tau is a special problem, thought to be relevant in some cases of bvFTD. Increasing the removal of phosphate from tau might be good; sodium selenate does that. That's logical, but remember, there is a huge amount about such neurodegenerative diseases that we do not know.
A small trial of selenate in patients with Alzheimer's disease has yielded ambiguous and confusing results. This is discussed in the Introduction to the current article.
News story: Promising treatment for dementia -- Sodium selenate slows behavioural variant frontotemporal dementia -- second most common dementia in under 60s. (Science Daily (Monash University), May 5, 2022.)
The article, which is open access: A phase 1b open-label study of sodium selenate as a
disease-modifying treatment for possible behavioral variant frontotemporal dementia. (Lucy Vivash et al, Alzheimer's & Dementia: Translational Research & Clinical Interventions 8:e12299, May 5, 2022.)
A post about the role of tau in a disease other than Alzheimer's: Tau and ALS (February 19, 2022).
More selenium biology... Did selenium deficiency play a role in mass extinctions? (February 5, 2016)
More selenium... A step toward a practical thermoelectric converter: get the oxide out (October 11, 2021).
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Brain. It includes a list of brain-related posts.
June 22, 2022
Briefly noted... COVID and pets?
June 22, 2022
It's something we have been wondering about, and we now have an update, based on collecting data in Toronto. Bottom line... Household cats and dogs do get infected with COVID-19. They develop antibodies, and sometimes get sick; occasionally, but not often, they may die from COVID. They probably get COVID from their human companions. There is no evidence that they transmit it to humans, but that cannot be excluded. The article also notes the issues of COVID in minks and deer; these are more serious.
* News story: Pets living in households with COVID-19 are high [sic] likely to seroconvert and become ill. (Neha Mathur, News-Medical.net, May 3, 2022.) Links to the article, which is open access.
* Direct link to the article, which is open access: Risk Factors for SARS-CoV-2 Infection and Illness in Cats and Dogs. (Dorothee Bienzle et al, Emerging Infectious Diseases 28:1154, June 2022.)
* There is a BITN section for SARS, MERS (coronaviruses). It includes a list of Musings posts in the field.
Silicates and gene expression -- a new way to induce making cartilage?
June 21, 2022
The following figure shows an effect of a silicate material on stem cells in lab culture...
The cells were stained for the enzyme alkaline phosphatase, after 14 days.
You can see that the cells grown with "nanosilicates" are different: they have more of the enzyme.
The other parts are for various ions from the material. They generally gave a response, too, especially the one with Si alone (as a soluble silicate).
This is part of Figure 2B from the article. The full Fig 2B also includes quantitative results from an enzyme assay for the same enzyme.
|
Here is more. More proteins responding to the nanosilicates (and its ions)...
In each case, more protein was made in response to the nanosilicates. The left-hand graph is for a form of collagen associated with making cartilage; the effect is about 15-fold in this case.
The quantitative results shown here are based on measuring bands on Western blots, normalized to a "standard" protein (β-actin).
This is part of Figure 2C from the article. The full Fig 2C contains data for other proteins, and also images of the Western blots.
|
What is this nanosilicate stuff? Many natural minerals are silicates. The nanosilicates used here are similar. The special feature is that the particles are small enough that they are readily taken up by cells. Then, they dissolve in the cell, releasing ions. The silicate ion may be the most important, but there are metal counter-ions in it, and they may also be relevant. (In the article, the Si product is described as Si(OH)4; don't take that literally.) (The evidence on the metal ions is mixed.)
What is the silicate material doing? It is affecting gene function, with the result of driving the stem cells toward being chondrocytes (cartilage). The figures above are pieces of the evidence. The proteins noted are important for those cells.
Why is that interesting? At one level, this is an exploration of how inorganic substances can control gene expression and, therefore, cell fate. In addition, the authors think this might be useful -- that using inorganic triggers might have advantages over using the more conventional growth factors (proteins). That, of course, is for future work to test.
News story: Minerals can be key to healing damaged tissue. (Nanowerk News (Texas A&M Engineering), May 6, 2022.)
The article, which is open access: Dissociation of nanosilicates induces downstream endochondral differentiation gene expression program. (Anna M Brokesh et al, Science Advances 8:eabl9404, April 27, 2022.)
Posts about cartilage include:
* Making new cartilage, using stem cells (January 5, 2022).
* Humans may be more like salamanders than we had thought (limb regeneration) (February 11, 2020).
Biology-related posts about silicon include:
* Miller-Urey revisited: the role of the glass container (January 22, 2022).
* Carbon-silicon bonds: the first from biology (January 27, 2017). Links to more about silicon.
There is more about stem cells on my page Biotechnology in the News (BITN) for Cloning and stem cells. It includes an extensive list of related Musings posts.
A connection between eating fish and skin cancer?
June 20, 2022
Computer databases are full of information about us. They can be queried to look for possible relationships between various factors. We now have an article asking whether there is a relationship between eating fish and getting melanoma (a form of skin cancer).
The following table shows part of the top-level analysis...

|
People were divided into five groups, based on how much fish they reported eating. The second column shows the average daily fish consumption for each quintile.
The next column shows the number of cases of malignant melanoma for each group. You can see that there is an upward trend: more fish correlates with more melanoma.
The final column shows the HR (hazard ratio), with 95% confidence limits, for each quintile, after some statistical adjustments. The first group (lowest fish consumption) is set to 1, as the reference value. The groups with higher fish consumption all showed about a 20% higher risk of malignant melanoma.
This is the left side (first analysis) of Table 2 from the article.
|
That's a correlation. The result says nothing about why this might happen. It is simply a lead for further work.
The next figure shows further analyses, each with only certain kinds of fish consumption considered...
This figure has three small tables. Each is similar to the one above, but for a subset of the data -- a subset of the fish.
The first table is for tuna consumption. It shows the same correlation. (The trend actually seems more consistent.)
The next two tables are for fried and non-fried fish. The correlation for fried fish is negative; that is, eating fried fish correlates with reduced levels of melanoma. The correlation for non-fried fish is positive.
This is the left side of Table 3 from the article.
|
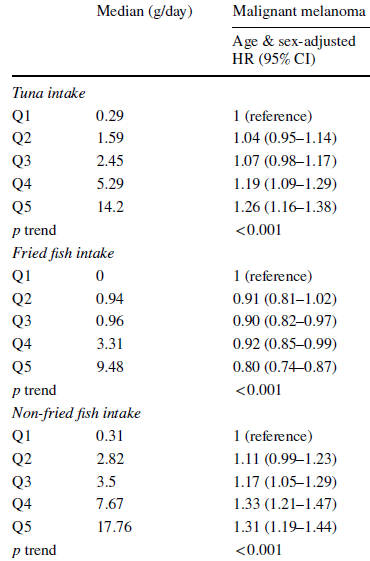
|
What does this all mean? I don't know, and the authors really don't either. (One possibility is that the results are due to environmental contaminants in fish.) They asked a question, and got an interesting and complex answer.
The authors note that there have been previous attempts to address the overall question of fish and melanoma, with widely varying results. The current study is the biggest yet. The second table suggests that we need to ask more specific questions. The effect of fish may depend on the type of fish and how it is prepared. It may also depend on where it comes from.
That's progress. We learn to ask better questions.
Other kinds of cancer? The authors note that previous analyses of the full data set showed an effect of fish on cancer only for melanoma.
News stories:
* Large Study Found a Strange Link Between Eating Fish And Skin Cancer. (Tessa Koumoundouros, Science Alert, June 11, 2022.)
* A novel study hints at a surprising link between eating fish and skin cancer -- The study followed 491,367 Americans aged between 50-71 to detect a link between skin cancer and fish consumption. (Loukia Papadopoulos, Interesting Engineering, June 12, 2022.)
* Could eating more fish raise your melanoma risk? - Expert Reaction. (Science Media Centre (New Zealand), June 10, 2022.) Links to more at other SMC sites.
The article: Fish intake and risk of melanoma in the NIH.AARP diet and health study. (Yufei Li et al, Cancer Causes & Control 33:921, July 2022.)
Among posts about melanoma: Anti-oxidants and cancer? (October 18, 2015).
Among posts about eating fish:
* Can an insect catch fish for its dinner? (October 8, 2018).
* DNA evidence in restaurants: is the fish properly labeled? (June 5, 2017).
* Omega-3 fatty acids; fish oil (March 29, 2010).
My page Internet resources: Biology - Miscellaneous contains a section on Nutrition; food and drug safety.
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Cancer. It includes an extensive list of relevant Musings posts.
How water droplets damage hard surfaces
June 18, 2022
The figure, from a recent article, shows how falling water droplets can damage a surface...
The structure in part a (left) consisted of tiny glass beads (about 90 micrometers diameter).
The structure in part b (right) consisted of a piece of plaster.
The structures were damaged by falling water drops. The nature of the drops was similar in the two cases.
The damage in a was done by a single water drop.
The damage in b was done by 2500 water drops.
The scale bars are 5 millimeters.
This is Figure 1 from the article.
|
The result in a is expected. But what's going on in b?
Here is some high speed photography....
Each row is a sequence of images showing what happened after a single drop hit the plaster surface.
The plaster contained a fluorescent marker, which could be tracked photographically. That allowed the scientists to measure the deformation in the material. The camera took 40,000 frames per second.
The top row is for a hard drop (a steel ball). The bottom row is for a water drop. (The size and speed of the drops were similar.)
The bright region shows where the shear stress is. What's important is the different pattern in the two rows. With the soft drop, the region of maximum stress moves along the material over time. With the hard drop, the region of maximum stress stays at about the same position.
The scale bar is 1 mm.
This is part of Figure 3 from the article.
|
The development of the high speed analysis was the heart of the current work. It's more than just optics. The measurements include shear stress and pressure in the object as a function of position and time. That lets them see what is effectively a shock wave that moves through the object when a soft drop impacts a hard surface. The results are fairly preliminary, but the scientists have developed a useful tool.
They call their method "high-speed stress microscopy".
News stories:
* Scientists find out why dripping water hollows out stone. (Stone-Ideas, April 7, 2022.) The site is about the architectural uses of stone, an area where erosion caused by water is an issue.
* New study solves mystery of how soft liquid droplets erode hard surfaces. (University of Minnesota, March 31, 2022.)
The article, which is open access: Stress distribution and surface shock wave of drop impact. (Ting-Pi Sun et al, Nature Communications 13:1703, March 31, 2022.)
Among posts about liquid drops...
* What would raindrops be like on other planets? (April 13, 2021).
* Speech droplets: Can you transmit an infection to someone by yelling "Stay healthy" at them? (June 14, 2020).
* Disease transmission by sneezing -- in wheat (July 29, 2019).
* The aroma of rain (June 13, 2015). Perhaps rather related to current article.
June 15, 2022
Briefly noted... How rotifers silence transposons
June 15, 2022
Transposons (mobile genetic elements, or, casually, jumping genes) increase the genetic variability in an organism -- for better or worse. For bdelloid rotifers they could be especially detrimental; these organisms lack sexual reproduction, which helps clean up the genome. In fact, rotifers have a relatively low content of transposons. A recent article shows that they have a novel way to keep transposons silent, based on an enzyme apparently acquired from bacteria by horizontal gene transfer.
* News story: New DNA modification system discovered in animals, captured from bacteria more than 60 MYA. (Science Daily (Marine Biological Laboratory), February 28, 2022.) Links to the article, which is open access.
* Direct link to the article, which is open access: Bacterial N4-methylcytosine as an epigenetic mark in eukaryotic DNA. (Fernando Rodriguez et al, Nature Communications 13:1072, February 28, 2022.)
* Why bring this up? Well, it is interesting science. But also, we note it to mark Transposon Day, which is tomorrow. Transposon Day, not surprisingly, is on the birthday of ...
- Transposon Day. This page is for 2021. I don't see a page for 2022. I wonder...
* Background post about rotifers: Lesbian necrophiliacs (March 8, 2010). Includes the discovery of HGT in rotifers; "paper 2" of that post is reference 16 of the current article. Includes music.
Male mice are stressed by the odor of bananas or pregnant females
June 14, 2022
It's a complicated story, but fun and perhaps even important. Let's just jump in and look at some of the results. We'll put the story together after a couple of figures.
The basic experimental tool here is a test of response to a pain stimulus. A defined pain stimulus (heat) is given to one paw; the time it takes for the mouse to withdraw the paw is measured. A slow response (compared to control conditions), may reflect stress. The test conditions all involve olfactory (odor) exposures.
The following figure establishes the basic phenomena...

The y-axis shows the response. It is labeled as the change (Δ) in latency: how fast (in seconds) the mouse responds to the pain stimulus under the specific conditions compared to control conditions. The measurements are for various conditions, listed along the x-axis. Part C (left) is for male mice; part D (right) is for female mice. The set of conditions is the same for both.
Messy data; each point is for one mouse, and they vary. To start, you can look at the marked central measures: the mean (big horizontal line) and its error bar. Even easier, you can just look across the top for asterisks, indicating that the result tests as statistically significant. The more *, the higher the statistical significance.
The most significant results are for two treatments with the male mice. These are shown with *** in red at the top. Male mice, exposed to female mice either in late pregnancy or lactating, with pups present. That is, males exposed to those females respond poorly to a pain stimulus. That condition is called pain analgesia.
There are three conditions with one *; we'll skip those, except to note that two are for small effects in the opposite direction.
This is part of Figure 1 from the article.
|
Another test, with the male mice...
Part B (left) shows the results for several specific chemicals. These are volatile chemicals; the test was done simply by exposing the male mice to the odor.
Three of the chemicals tested gave a ** or *** result.
Part C (right) shows a test using banana oil, from the grocery store. It gave a strong response (right side). (The left side of Part C shows the results for the individual mice: latency before and after the treatment.)
Female mice showed no response to these chemicals.
This is part of Figure 4 from the article.
|
The story? Male mice are a threat to females with pups. That is well-known. They may even kill the pups. Females act to protect the pups.
The first figure here shows this behavior, translated into a quantitative test that can be used in the lab. The male mice show a behavioral change, pain analgesia, in response to females near birth (late-pregnant or lactating).
Analysis of the urine from female mice at various stages showed that several chemicals were present at high levels during that same time. That is, the presence of these chemicals in the female's urine correlated with the resulting male behavior.
The second figure above shows a test of those chemicals, alone. Some of them are active in promoting the response. That is, not only do these chemicals correlate with male behavior, but they cause it.
One of those chemicals -- the most active one, perhaps -- is very similar to the major odor in bananas. n-Pentyl (amyl) acetate in the urine, isopentyl (isoamyl) acetate in bananas. That led the scientists to test banana oil; it worked -- very well. Is there any significance to the banana result? Not obvious, but it is fun to note it.
The big story here is working out how female mice protect their young from competing males. That involved developing a lab test, and identifying specific chemicals involved in repelling the males.
The work is relevant to the use of mice in lab work. In recent years people have become aware that the way lab animals are treated can affect the experimental results. An earlier post about this is noted below.
News stories:
* A coincidental finding shows that male mice are afraid of bananas. Here's why -- A compound called n-pentyl acetate has something to do with it. (Mert Erdemir, Interesting Engineering, May 30, 2022.)
* Why are male mice afraid of bananas? (Phys.org (McGill University), May 24, 2022.)
The article, which is open access: Olfactory exposure to late-pregnant and lactating mice causes stress-induced analgesia in male mice. (Sarah F Rosen et al, Science Advances 8:eabi9366, May 20, 2022.)
Posts about mice behavior include...
* Why male scientists may have trouble doing good science: the mice don't like how they smell (August 22, 2014).
* Would wild mice use an exercise wheel? (July 11, 2014).
Posts about bananas, including the odor, include...
* Food security: the potential of enhanced cultivation of enset (February 22, 2022).
* What does blue light smell like? (July 18, 2010).
Graphdiyne: helping make silver a better anti-bacterial agent
June 11, 2022
Graphdiyne? It's a variation of graphene, but with some triple bonds. A new article reports an interesting use of graphdiyne, to stabilize silver so that it is more effective as an anti-bacterial agent.
More about the nature of graphdiyne towards the end, but that is not needed to follow the main story of the current article.
Here are some results...
Three agents were tested against two types of bacteria.
The graphs show the MIC for each agent. MIC = minimum inhibitory concentration. The lower the MIC, the more potent is the agent.
The bacteria are Bacillus subtilis (left side) and Escherichia coli (right), taken as representative of gram-positive and gram-negative bacteria, respectively.
The three agents are, starting at the right: AgNPs (silver nanoparticles), GDY (graphdiyne), and GDY@Ag (graphdiyne with Ag in it).
In each case, the MIC was much lower for the new combined agent, GDY@Ag. (Note that the y-axis scale is split, just below the 60. So the difference is greater than it appears visually.)
In particular, the GDY@Ag complex was more effective than the silver alone. The GDY alone had no activity, as judged by these tests.
This is part of Figure 3 from the article.
|
The following figure shows the effect of the combined agent as judged by growth curves.

The graphs show bacterial growth vs time. Bacterial growth was measured by OD600, the optical density at 600 nm.
In each case, the light gray curve (at the left) shows the growth of the control culture, with no drug added.
The red curve shows the growth when GDY@Ag was added at the MIC level (as shown in the first figure). In both cases, that level led to essentially no growth over the time period studied.
The middle curve (darker gray) shows the growth at a level of the agent that was half the MIC level. In both cases, that level delayed growth, compared to the control curve. The growth delay was more for the E coli bacteria (right-hand graph). However, once growth started, it was fairly normal, both for rate and final amount of bacteria.
Note that the x-axis scale is mixed linear and log, with no marking to show the change. Most important for us is the first part of the graphs, in the linear-scale region (0-18 h).
This is part of Figure 4 from the article.
|
What is this combined agent GDY@Ag? Rather simple, actually. They just mix silver nitrate, as a source of silver ions, with the graphdiyne. The GDY reduces the Ag+ to elemental Ag, which is contained within the GDY structure.
Why is GDY@Ag better than Ag alone? There is much consideration of the use of Ag as an anti-bacterial agent. But it is hard to use. It may be that ordinary Ag preparations lose their effectiveness as the Ag aggregates into larger particles. The GDY@Ag may be good precisely because it holds the Ag at multiple sites throughout the GDY, thus limiting the ability of the Ag to aggregate further.
Overall, the work is an interesting development in the often-murky story of the use of Ag as an anti-bacterial agent, and is also an exploration of a new form of carbon.
News story: Graphdiyne functionalized by silver nanoparticles to combat the threat of antibacterial resistance. (Yashwant Mahajan, Nanowerk, May 11, 2022.)
The article, which is open access: Insight into the antibacterial resistance of graphdiyne functionalized by silver nanoparticles. (Simin Qin et al, Cell Proliferation 55:e13236, May 2022.)
A bit about graphdiyne...

The figure shows graphene (at the right), graphdiyne (the major structure shown, in yellow), and part of a carbon nanotube (left, in black; not labeled here). It's an artist's view, showing the basic geometry. It omits some chemical details. For example, the alternating double and single bonds in the rings are not shown, nor are the triple bonds of the linker.
The linker is based on butadiyne: H-C≡C-C≡C-H. That molecule is linear; the C atoms are all sp-hybridized. The linker itself is that without the H; that is, it is linear, and includes two triple bonds (di-yne). Graphene itself is planar; adding a linear linker such as this does not change that.
The structure of graphdiyne may give you some ideas. What if there were only one triple bond? That would be graphyne -- another material, but not the immediate subject.
In the graphdiyne structure shown above, note the large "empty" triangular regions. It is likely that the Ag atoms are deposited in those regions.
This is part of a figure accompanying some versions of the following article: Architecture of graphdiyne nanoscale films. (Guoxing Li et al, Chemical Communications 46:3256, 2010.) The figure is not in the original article, but is part of the pdf of the article from the authors. Check Google Scholar for a freely available copy. The following link is one: pdf, with the full version of this figure as the first page. The full figure has more structures!
|
My page for Biotechnology in the News (BITN) -- Other topics includes a section on Antibiotics. It includes a list of Musings posts on the topic.
Posts about graphene and related structures are listed on my page Introduction to Organic and Biochemistry -- Internet resources in the section on Aromatic compounds.
June 8, 2022
Briefly noted... Can a fish drive a truck?
June 8, 2022
Humans can navigate under water. Can fish navigate on land? And will they seek rewards there? A recent article addresses those questions. It's a fun story, but also some good science about animal behavior.
* News stories. Both link to the article.
- Scientists taught a goldfish to drive a watery tank on wheels - and it's pretty good at it. (Fermin Koop, ZME Science, January 12, 2022.)
- Goldfish driving 'cars' offer new insight into navigation. (Maria Temming, illustrated by JoAnna Wendel, Science News for Students (now Science News Explores), February 16, 2022.) Presented in part as a comic strip.
* Direct link to the article: From fish out of water to new insights on navigation mechanisms in animals. (Shachar Givon et al, Behavioural Brain Research 419:113711, February 15, 2022.) Check Google Scholar for a freely available copy from the authors.
Encoding peptides from primordial RNA?
June 6, 2022
A common view of how life got started is expressed in the RNA World model. RNA can both carry genetic information and do catalysis; it seems likely that RNA was the key player in the early stages of life.
In the modern world, nucleic acid (RNA or DNA) codes for protein, which does (most of) the catalysis. How did that transition happen? A new article provides evidence for a simple form of RNA-encodes-protein.
Here's the idea...
Part a (left) shows how modern protein synthesis occurs. Briefly, an amino acid is transferred to the end of a growing peptide chain. Both the incoming amino acid and the growing chain are attached to RNA molecules, specifically transfer RNA (tRNA) molecules. Both tRNA molecules are paired to the messenger RNA (mRNA) molecule.
Part b (right) shows the scheme proposed in the current work. Briefly, an amino acid is transferred to the end of a growing peptide chain. Both the incoming amino acid and the growing chain are attached to RNA molecules. The two RNA molecules, carrying the incoming amino acid and the growing chain, are paired to each other.
This is Figure 1 from the news story (by Bonfio) accompanying the article.
|
Those two processes are rather similar -- so long as there is a requirement that the incoming amino acid's RNA actually pairs properly with the RNA of the growing chain. That is the feature that can lead to encoding.
Here is an example of what they actually did...

|
Start at the upper left. There are two amino acids (G & V -- glycine and valine), each attached to a small RNA molecule (blue, red). Those two RNAs are paired, in the usual way.
The details are such that the two amino acids react with each other, forming a peptide (amide) bond. That step is labeled 'coupling'.
The top amino acid (G) is cleaved from its RNA (and the RNA is removed). This exposes the -NH2 group of the G. That step is labeled 'cleavage'. We now have a dipeptide, still attached to the lower RNA.
|
The cycle is now repeated in the right-hand column, adding another G to the growing chain.
This is part of Figure 3b from the article. The full Figure 3b shows more cycles. The first three parts can be considered a sequence (the two parts shown above, plus one more). The last two parts can be taken as examples.
|
What's behind this scheme? It is quite likely that amino acids and small peptides were around during the RNA World stage. They may have even been abundant. And they may have been attached to RNAs -- just as they are in the modern world. Suggesting that such hybrid molecules were around got the scientists thinking how that might lead to a coding system. The current system begins to show how that might work.
As always, distinguish what they have shown from any claim that this is what happened. The point of the current article is that it gets us thinking about the problem of how nucleic-acid-codes-for-protein might have developed, and it offers a specific example.
Notes...
The current work includes an experiment testing the length of the paired region that is needed. Interestingly, it is about three bases. Is that related to the length of the modern codon, or just a coincidence?
The direction of chain growth here is opposite to modern protein synthesis.
News stories:
* The origin of life: A paradigm shift. (Science Daily (Ludwig-Maximilians-Universität München), May 12, 2022.)
* A pair of experiments reveal new details regarding the origin of life on Earth. (Philip Guelpa, World Socialist Web Site, June 2, 2022.) Interesting source. But it is a useful presentation of the work. As the tittle suggests, it actually deals with two articles. One is the article of a recent (March 26) post listed below.
* News story accompanying the article: Origins of life: A possible path towards encoded protein synthesis -- How did the biological machinery for protein synthesis evolve from simple chemicals on ancient Earth? Experiments suggest an intriguing role for modified RNA nucleotides in directing stepwise peptide synthesis. (Claudia Bonfio, Nature 605:231, May 12, 2022.)
* The article, which is open access: A prebiotically plausible scenario of an RNA-peptide world. (Felix Müller et al, Nature 605:279, May 12, 2022.)
Added October 22, 2025.
More about the prebiotic synthesis of proteins: Role of thioesters in prebiotic RNA-based protein synthesis? (October 22, 2025).
A recent post about early life... An RNA replicator network (March 26, 2022).
Another unusual way to make protein... Making peptides in space: a new pathway (April 15, 2022).
My page Internet Resources for Organic and Biochemistry has a section on Amino acids, proteins, genes. It includes a list of posts about making proteins.
Acholetin: a new polymer from bacteria
June 4, 2022
A recent article reports a new polymer. There are various reasons it might be interesting, but we first need to establish what it is.
The following figure gives some basics, on two issues. Both parts are fairly complex, but we'll focus here on some key points.

Let's start with Part B (bottom). The molecule at the left (blue) is based on glucose, with two modifications. First, it has a phosphate group, at position 1; this activates the sugar, so it can join with others. Second, it has an amino group, at position 2; actually, it is an N-acetyl amino group. This 2-N-acetylamino is found in chitin; more about that later.
The next structure (black) is a polymer, built from the first molecule. The n at the lower right of the big brackets indicates that the bracketed unit is repeated n times. What's important is the linkage, which is between the 1-position of the left-hand sugar and the 3-position of the right-hand sugar. 1,3. Actually, it is β-1,3, where the beta describes the orientation.
To the right of the arrow... Those first two molecules are now joined. Again, 1,3. The polymer is one sugar longer than before. And a phosphate was released.
The arrow is labeled AchP. That's the enzyme, which was discovered in this work. The arrow is bi-directional. That same enzyme catalyzes both the polymerization and depolymerization.
Overall, part B describes the basic claim: a polymer of 2-(N-acetylamino)glucose linked β-1,3. That is a new polymer. Further, they have the enzyme that can both make and degrade the polymer.
Part A (top) shows some of the properties of the enzyme, in lab tests. The basic plan was to test various donor and acceptor molecules to see if the joining occurred. The donor is the one with the phosphate; the acceptor is the growing chain. The properties are fairly complex; we'll just note some highlights.
Three donors were tested. They all have that 1-phosphate group. The first two rows are donors based on glucose (Glc); the bottom row is for a donor based on galactose (Gal). The color of the boxes shows how good the reaction was. The Gal donor did very little. The two Glc donors worked, but the one with the N-acetylamino (NAc) worked best.
Several acceptors were tested. Let's just look at the first three for now. Glucose, aminoglucose, and N-acetylaminoglucose. The last of those worked best, but the full picture is more complicated. Among the other acceptors tested, it is interesting to note that the Gal-based acceptors worked rather well, too.
The numbering of the positions on the sugar is standard, and is consistent. I have added some numbers on the molecule at the left. The phosphate is at the 1-position. The N (in this case with an acetyl group) is at 2. I added numbers for the -OH at 3, 4, and 6. The numbers should be on the C atoms, but I put them where there was some space. In the larger unit to the right, you can see that the linkage between sugars is 1,3. It is beta, due to the orientation.
This is part of Figure 2 from the article.
|
That's a long discussion of (part of) a single figure! In brief...
- We have a novel polymer.
- It's from nature, from bacteria.
- We have the enzyme to make it. The enzyme properties are consistent with the polymer found, but also suggest there could be additional complexity.
- The enzyme can also degrade the polymer.
The discovery was rather accidental. The scientists were screening genome databases for enzymes that might degrade chitin. Of course, enzymes that work on things similar to chitin might pop up.
The bacterial source here is an unusual one, without a cell wall. It is Acholeplasma laidlawii. Of course, database screens require only that a genome has been deposited, and are not biased by the nature of the organism.
As to the polymer... There are three ways to connect two molecules of the starting sugar in the β orientation: 1,3; 1,4; and 1,6. See the right hand molecule in part B, above. The 1,4 version is the familiar polymer chitin. The 1,6 version is also known. Thus finding the 1,3 version completes the set.
The new polymer has been given the name acholetin, reflecting the name of the bacteria and the connection to chitin. The enzyme is called acholetin phosphorylase (AchP).
Is the polymer (or the enzyme) of practical use? That remains to be seen.
Are there other molecules out there in the natural world still waiting to be discovered? Probably.
News stories:
* Bacterial Enzyme Makes New Type of Biocompatible, Biodegradable Polymer. (SciTechDaily (American Chemical Society), March 16, 2022.)
* Bacterial enzyme produces biodegradable polymer. (Nanowerk News (Advanced Light Source, Lawrence Berkeley National Laboratory), May 21, 2022.)
The article, which is open access: A Synthetic Gene Library Yields a Previously Unknown Glycoside Phosphorylase That Degrades and Assembles Poly-β-1,3-GlcNAc, Completing the Suite of β-Linked GlcNAc Polysaccharides. (Spencer S Macdonald et al, ACS Central Science 8:430, April 27, 2022.)
More about chitin, and its derivative, chitosan: Some shrimp in your wine? (August 27, 2016).
My page Organic/Biochemistry Internet resources has a section on Carbohydrates. It includes a list of related Musings posts.
June 1, 2022
Briefly noted... Do cats know the names of their house-mates?
June 1, 2022
Cats recognize their names. Do they also recognize the names of other cats they live with? To test that, scientists tested how the cats responded to seeing a picture of one of their cat-friends on the computer screen, along with hearing a name that either did or did not match. Housecats, who would be familiar with hearing the names of their cat-friends, showed a small difference in how they responded to a match vs mismatch. Cats from an environment where they were not exposed to the names did not show such an effect. It's hard to know what to make of this. The idea is interesting, and the experiments clever. But the results are hard to interpret. There are some hints, but no big conclusions. One wonders what the follow-up will be. Apparently one of the subjects didn't think much of the work, and simply left after the first trial.
* News stories. Both link to the article, which is open access.
- Cats learn the names of their feline friends. (Big Think, May 25, 2022.)
- Cats can remember each other's names, study says. (Chris Melore, Study Finds, May 16, 2022.)
* Direct link to the article, which is open access: Cats learn the names of their friend cats in their daily lives. (Saho Takagi et al, Scientific Reports 12:6155, April 13, 2022.)
Added September 9, 2025.
More about cat behavior: The genetics of cat purring (September 9, 2025).
How many exoplanets are known? Three (four?) fewer than we thought
May 31, 2022
Recent years have seen the discovery of thousands of exoplanets -- planets beyond our Solar System. Musings has noted some of the investigations, including the methodology for recognizing a planet at a distance [links at the end].
A recent article re-examines some of the identifications, and argues that at least three objects previously reported by the Kepler mission as planets are, more likely, stars.
The following figure shows the key evidence. The issue is that the reported sizes of some planets are in doubt. That is because the sizes of their host stars are in doubt, and the reported planet size is directly related to the size of the host star.

|
The graph plots the apparent size of a collection of exoplanets -- based on two different catalogs (KIC & TIC) that give the sizes of the host stars. The green diagonal line shows what would be expected if the catalogs agreed.
Planet size is shown as RJup. That is radius, relative to Jupiter.
Most of the planets fall fairly close to the diagonal line.
|
However, four are particularly noteworthy; they are shown in red. These four are all above the line; they are larger when judged by the TIC than by the KIC. The KIC was used for the original classification. The TIC is based on newer and better data. (In particular, there is better data for the distance of each star from Earth, which is used to find the size -- of the star, and hence of the planet.) The graph, then, suggests that these planets might be considerably larger than expected during the original analysis. And they are big -- 2-3 times the size of Jupiter (by radius), if the larger number is correct.
KIC = Kepler Input Catalog; TIC = TESS Input Catalog.
This is the lower left part of Figure 1 from the article.
|
The basic story is about that simple. (There is considerable additional detail, to check that other known features of the system fit.) Remember, a factor of 2 in radius gives a factor of 8 in volume. The biggest planet candidates are just too big to be planets, now that we have better data on their sizes.
The work actually began with a different purpose. The scientists wanted to examine the shape distortion that can occur when two massive objects are near each other. Of course, they started with planets that seemed rather large. But the results showed effects that were considerably larger than expected.
That led them to re-examine the reported sizes. Turns out that the measurements of star size have improved. Kepler may seem to be a very recent mission, but star measurements have improved since then. The TIC is based on the European Space Agency's Gaia mission, launched only in 2013. (Kepler was launched in 2009, and did most of its successful work by 2013.)
For the smallest of this set of four suspects, the size per se is marginal, but other factors suggest to the scientists that it may well not be a planet.
News story: Look! Up in the sky! Is it a planet? Nope, just a star. (Science Daily (Massachusetts Institute of Technology), March 15, 2022.)
The article, which is open access: Revisiting Kepler Transiting Systems: Unvetting Planets and Constraining Relationships among Harmonics in Phase Curves. (Prajwal Niraula et al, Astronomical Journal 163:172, April 2022.)
Background posts about exoplanets include...
* A new trick for the Kepler planet-hunters (June 25, 2012). The post includes a prediction "that over 90% of Kepler candidates will be validated as planets." Losing 3 or 4 is not a big deal. What is important about the new article is the improved characterization.
* The Kepler Orrery (June 3, 2011). Links to more.
Sleep genes and Alzheimer's disease?
May 28, 2022
It is known that Alzheimer's disease (AD) is associated with poor sleep.
It is also known that there are some people who thrive on a relatively small amount of sleep. In some cases, the trait runs in families, and is called FNSS = Familial Natural Short Sleep. FNSS has been associated with mutations in at least two genes. So far, it seems that people with FNSS do not have high levels of neurodegeneration, such as AD. That is, such people sleep less, but do not have the common correlates of reduced sleep.
A new article provides evidence, in a mouse model of AD, suggesting that those with FNSS mutations have reduced development of some AD features.
Here are the results from one test...

This test is about tau, a protein associated with AD (and with other neurodegenerative conditions). The measurement is for the amount of a particular form of tau (phosphorylated tau) that is associated with AD. It is measured with an antibody that is specific for that form of tau. Green indicates a response.
Part A shows representative images of what was found for three mouse strains; part B (right) shows the quantitative results, over the multiple mice examined.
PS19 (at the left) is the basic mouse strain used as the AD model. (It carries human genes that lead to AD.) DEC2 is one of the genes that can lead to FNSS. The middle image is for the WT (wild type) form of the gene. The right-hand image is for P384R, a mutant form of the DEC2 gene, leading to short sleep.
Visual inspection of Part A suggests that the mouse with the 'short sleep' mutation has less phosphorylated tau. Part B confirms that there is a significant effect of the mutation on reducing the amount of phosphorylated tau. We also note that the distributions in part B are not simple; there is a hint that the effect of the mutation is to reduce outliers at the high end.
This is part of Figure 1 from the article.
|
That's tau. Another protein associated with AD is the amyloid beta peptide, Aβ42. The following figure shows some results for that AD-related protein...

|
The first two bars (left; gray and blue) are for the same two DEC alleles used in the first test, in mice at age three months. The results show the amount of Aβ42 in one brain region. They are very different. This is the key result from this figure.
The right side of the figure shows results for mice at six months. Look at the same color bars (gray and blue) there, and there is no significant difference. Again, the eye may suggest otherwise.
|
The other two bars (black and red) show results for wild type and mutant alleles of another 'short sleep' gene tested. Although there is no significant effect in this test, that mutation did have other effects on Aβ42.
In this test, 5XFAD is the mouse strain used as the AD model.
This is Figure 3B from the article.
|
Overall, both FNSS mutations tested, in different genes, affected AD markers, but differently.
What does this all mean? As so often with AD, that is not clear. At a minimum, the work offers some clues about the connection between sleep and AD. Two genes first described as affecting sleep also affect AD proteins -- at least in a mouse model.
"This study employs an understudied approach for AD (both Aβ and tau) by investigating strong genetic factors that have potential in conferring resistance to onset and progression of AD-like pathology. This work was motivated by our idea that natural short sleepers are accomplishing restorative aspects of sleep more efficiently. The results indicate that there is great potential in identifying the means to use improved sleep as a target for protecting against neurodegeneration, thus decreasing the prevalence of AD, and potentially, other forms of neurodegeneration." (From the Discussion section of the article, near the end (p 10 of the pdf).)
The 'short sleep' genes allow people to thrive on a small amount of sleep. That would seem to be a beneficial trait. However, the trait is rare. Why? Are we missing something important about what these mutant alleles are doing? In any case, study of FNSS and its genetics should continue to be of interest. (The main research teams of this work are sleep researchers. They used AD as a tool in their study of sleep.)
There is more to be done.
News stories:
* Genes May Decide How Much Sleep We Need. (Dylan Roche, Sleep Foundation, April 8, 2022. Now archived.)
* Genes for shorter sleep linked with reduction of Alzheimer's changes in mice brain. (Alzheimer's Research UK, March 16, 2022.)
* When it comes to sleep, it's quality over quantity. (Science Daily (University of California - San Francisco), March 15, 2022.)
The article, which is open access: Familial natural short sleep mutations reduce Alzheimer pathology in mice. (Qing Dong et al, iScience 25:103964, April 15, 2022.)
A recent post about sleep, including the role of a mutation: What happens to capillary blood flow in the brain during sleep -- and why? (November 8, 2021).
More sleep...
* Is it worthwhile to sleep for four seconds? (February 20, 2024).
* A gene for insomnia? (February 4, 2023).
A recent post about AD: How many types of Alzheimer's disease are there? (June 15, 2021).
My page for Biotechnology in the News (BITN) -- Other topics includes sections on Alzheimer's disease and Brain. Each includes a list of related Musings posts. The 'brain' section includes many posts on sleep.
May 25, 2022
Briefly noted... How useful is it to emphasize a "natural" approach to removing CO2 from the air?
May 25, 2022
One way to reduce atmospheric carbon dioxide is to promote its use by plants. That can be thought of as a nature-based, or "natural", approach to CO2 reduction. A concern about that approach is that it could be temporary; CO2 in plants can have various fates; one of them is to be returned to the atmosphere. A recent article analyzes the approach, and how it interacts with other approaches. The take-home lesson is that incorporation of CO2 into plants is good. However, it must be done in conjunction with other approaches to reduce CO2 for a robust long-term effect. That balance is probably not a surprise, but the article provides good analysis.
* News stories. Both link to the article, which is open access.
- Nature-based carbon removal can help protect us from a warming planet. (Nanowerk News (Simon Fraser University), March 29, 2022.)
- Planting trees can help the climate, but only if we also stop burning fossil fuels. (The Conversation, March 30, 2022.) From three of the authors of the article.
Effect of the COVID pandemic on the incidence of dengue
May 24, 2022
During the COVID-19 pandemic there has been a major reduction of the incidence of influenza. Musings noted an early report of the effect [link at the end]; it has held up.
The incidence of dengue has also declined during the COVID pandemic. COVID and flu are both due to respiratory viruses; their modes of transmission are probably similar. But dengue is transmuted by mosquitoes; the connection between COVID and dengue may raise different issues. We now have an article exploring that connection.
Here is the basic phenomenon...
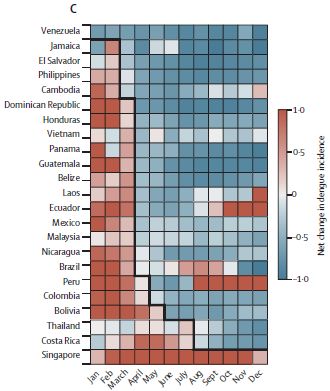
|
The figure shows the incidence of dengue in 23 countries (listed along the left side) during the months of 2020 (x-axis); these are all countries with a high incidence of dengue. As many will recall, COVID restrictions began around March 2020.
The monthly incidence is reported relative to the incidence in recent years for the same month. Red is for higher levels; blue is for lower levels.
The big picture is that most countries started the year with relatively high levels of dengue. After March, the rest of the year was largely marked by low levels of dengue.
The article notes that some countries with high levels of dengue were not included in the analysis here, because the necessary monthly data was not available.
This is Figure 1C from the article.
|
It is clear that a substantial reduction of dengue occurred during the time of COVID restrictions. (For 2020 as a whole, this set of countries showed about a 44% decline in dengue.)
The authors went on to analyze the nature of COVID restrictions, to see if they could identify any features that seemed of particular relevance to the reduction of dengue.
The analysis involved statistical modeling, using a variety of data, from various sources. The authors discuss the limitations of the analysis.
The following figure summarizes one part of the analysis...

|
The figure shows the two features of COVID restriction that showed the biggest effects in the analysis. The results are shown here for two geographic groups of countries.
This is the bottom part of Figure 4 from the article. (The rest of the figure shows the results for the individual countries. The big picture is that they are similar.)
|
As you can see there, the analysis suggests that the time spent away from home (in school or 'other') is a major factor.
The authors are not interested in restoring restrictions as a tool against dengue. Instead, they hope that their analysis might offer clues about where to focus anti-dengue efforts. For example, the results shown above might suggest that mosquito control should be focused on places where people congregate (including schools), rather than on the home.
News stories:
* Pandemic restrictions could be linked to 750,000 fewer dengue cases -- The insight could help us better manage the disease. (Fermin Koop, ZME Science, March 4, 2022.)
* COVID-19 restrictions linked to nearly 750,000 fewer dengue cases in 2020 -- New study shows COVID-19 measures such as school closures and 'high-traffic and mixing' areas had the strongest association, providing clues for new intervention approaches. (Science Daily (London School of Hygiene & Tropical Medicine), March 3, 2022.)
* "Comment" accompanying the article: Movement dynamics: reduced dengue cases during the COVID-19 pandemic. (R Tedjo Sasmono & Marsha S Santoso, Lancet Infectious Diseases 22:570, May 2022.) Caution... The authors are from Indonesia. They spend much of their Comment discussing the situation there. Only after presenting that in some detail do they reveal that the article analysis does not include Indonesia (a country that did not have the monthly data needed here). It's still an interesting story, but the way it is organized leads to a letdown.
* The article, which is open access: Measuring the effects of COVID-19-related disruption on dengue transmission in southeast Asia and Latin America: a statistical modelling study. (Yuyang Chen et al, Lancet Infectious Diseases 22:657, May 2022.)
Background post about influenza during the COVID pandemic: How bad will the upcoming COVID-era winter flu season be? (September 25, 2020).
My page for Biotechnology in the News (BITN) -- Other topics includes sections on Dengue virus (and miscellaneous flaviviruses) and SARS, MERS (coronaviruses). They include lists of related Musings posts.
What to do with silicon waste from dead solar cells
May 23, 2022
It's a sign of progress, I suppose. We've been using solar (photovoltaic) panels long enough that we are facing a new waste problem: what to do with dead solar cells.
A new article reports work on converting solar cell waste into thermoelectric (TE) generators, which make electricity from temperature (T) gradients.
A key insight was to realize that, while both can be silicon-based, they use the silicon in different ways. Specifically, the TE device is not affected by the impurities that convert bulk Si into useful solar cells. So, one can just take the waste solar panels, grind them, extract things that seem valuable, and then re-purpose the Si to the new task. It is not necessary to purify out the dopants used to make solar cells.
The following figure gives an idea of how the resulting TE devices perform...
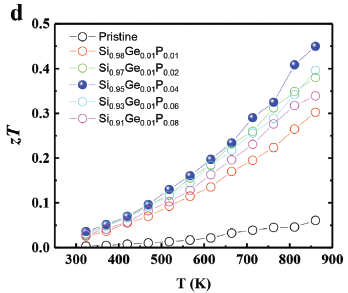
|
The graph shows zT vs T for various materials.
zT is a complex parameter, often taken as a single number reflecting the efficiency of the TE system; it is called the figure of merit.
TE devices are driven by a temperature difference. The T shown on the x-axis is the hot end. The cold end was constant, at 323 K.
The lowest curve is for the "pristine" silicon, as recovered from the solar cell waste. The other curves are for that material, with additions of germanium and phosphorus. The Ge is constant (at 1%), but P is varied. All are better than without additives, but there is an optimum at 4% P.
|
The germanium helps reduce the thermal conductivity. The phosphorus increases the electrical conductivity.
This is Figure 5d from the article.
|
The waste Si from the solar panels is now working in functioning TE devices.
These are not particularly good TE devices. TE still has limited use, due to poor performance. The highest zT obtained here is high for Si-based TE devices, but well below state-of-the-art for TE. On the other hand, these Si-based TE devices are rather light, which may be a significant advantage for some uses (such as on airplanes). Further, they may be relatively inexpensive, given the Si source.
Of course, further work may result in better Si-based TE devices.
In any case, the work represents a step toward figuring out what to do with a new waste material.
News story: Upcycling strategy turns discarded solar cells into thermoelectrics. (Michael Berger, Nanowerk Spotlight, May 16, 2022.)
The article: Upcycling Silicon Photovoltaic Waste into Thermoelectrics. (Jing Cao et al, Advanced Materials 34:2110518, May 12, 2022.)
A recent post about both solar cells and TE devices: A solar cell that generates electricity at night (April 12, 2022). The news story listed above mentions the work behind this recent post.
There is more about energy issues on my page Internet Resources for Organic and Biochemistry under Energy resources. It includes a list of some related Musings posts.
How to make bricks on Mars
May 21, 2022
At some point, we are going to need building materials on Mars.
Here are some Martian bricks, from a recent article...
Actually, they are pseudo-Martian bricks. Made on Earth, but by a process that suggests how one might make bricks on Mars.
They were made using Martian Simulant Soil (MSS), which one can buy from a company in Florida.
The major dimensions are probably about 3 centimeters, judging from the description of the apparatus. The process should be scaleable to any size desired.
The scientists had previously made Lunar bricks, using Lunar Simulant Soil (LSS), which apparently they had to make for themselves. Work with LSS continued here.
This is Figure 2 from the article.
|
How do you do that? The general idea is straightforward. There is soil on Mars. We know there are various kinds of soil, including some that is very hard; we even use the term brick-hard soil. The issue is, how do we take the soil we find, and convert it to something suitable for use as bricks.
The scientists try various things, including adding metal ions and polysaccharide gums. In particular, they use a bacterial process, with the bacterium Sporosarcina pasteurii.
How good are the bricks? The following figure shows some data. The graph shows the strength -- compression strength -- for samples of Martian brick made in different ways.

|
The bar labels all include MSS-SP: Martian Simulant Soil and the bacterium.
GG and N refer to guar gum and nickel ions, Ni2+, respectively.
You can see that the right-hand bar, for bricks made with all these additives, is the strongest.
Samples made with MSS-SP alone, with neither GG or N, were not strong enough to be tested.
This is Figure 5a from the article.
|
The compression strengths found here are not very high, but may be high enough for making useful temporary structures. Remember, the starting material (MSS) used here is loose soil.
What are the bacteria doing? The scientists think the key role is to make ammonia, NH3, thus raising the pH of the soil; that promotes precipitation of minerals such as calcite (calcium carbonate). (The bacteria used here make the enzyme urease, which makes NH3 from urea. The growth conditions include urea. The bacteria infiltrate the soil, and thus make NH3 throughout. The urease enzyme requires Ni2+.)
The work opens the door to making bricks on site on Mars. As noted earlier, they also made Lunar bricks, by more or less the same procedure. More generally, they call their results space bricks.
News stories:
* ISRO-IISc team develops prototype of bacteria-infused bricks for Martian, lunar soil -- New method is proof of concept for sustainable way to make bricks for construction on Mars & Moon using soil found on surface. (Sandhya Ramesh, The Print, April 22, 2022.)
* Using bacteria to build settlements on Mars. (Nanowerk News (Indian Institute of Science Bangalore), April 20, 2022.)
The article, which is open access: Microbial induced calcite precipitation can consolidate martian and lunar regolith simulants. (Rashmi Dikshit et al, PLoS ONE 17:e0266415, April 14, 2022.)
More about Mars...
* Could Deinococcus radiodurans survive on Mars? (November 6, 2022).
* Another underground lake on Mars -- near the equator? (April 5, 2022).
* Perchlorate on Mars surface, irradiated by UV, is toxic (July 21, 2017). This issue is not dealt with in the current article.
May 18, 2022
Briefly noted... The youngest known asteroids
May 18, 2022
Two asteroids were (independently) discovered "near" Earth in 2019. Study of them suggests that they are siblings -- and that they were born only about 300 years ago, That would make them the youngest known asteroids. It is likely that they originated from a comet. The data is limited at this point. Much of what is claimed is based on modeling, and much is unclear. More data should become available in 2033, when these baby asteroids will next be close enough for observation.
* News story: Astronomers Spot The Youngest Pair of Asteroids Ever Discovered in The Solar System. (Michelle Starr, Science Alert, February 7, 2022.)
* The article, which may be freely available: Recent formation and likely cometary activity of near-Earth asteroid pair 2019 PR2-2019 QR6. (Petr Fatka et al, Monthly Notices of the Royal Astronomical Society 510:6033, March 2022.)
A gold-based spray to treat heart damage
May 17, 2022
Heart tissue tends to die after a myocardial infarction (heart attack). A treatment that reduced that tissue death would be good.
A recent article reports development of a spray that might help. That raises a number of questions. Let's start with some evidence that it works -- in mice...

|
The mice were given an artificial (lab-induced) myocardial infarction. They were then given various treatments, and evaluated by several measures a month later.
The graph shows a measure of scarring.
|
The left-hand bar is a control, with a mock treatment (PBS = phosphate-buffered saline) The scarring is about 50%. The right-hand bar is for the preferred treatment. The scarring is reduced to about 10%, a major improvement.
That preferred treatment is labeled Multi-AuNP. The two components of that were tested individually, and gave some improvement. A couple of other things were tested; we'll skip them here.
This is Figure 4c from the article.
|
The treatment worked. It substantially reduced scarring. Several measures of structure and function were consistent on this point.
What is this Multi-AuNP stuff? AuNP stands for gold nanoparticles. Multi is a peptide that the scientists studied. (Leg-4 is another peptide; it didn't work as well.)
How is the treatment done? How do you spray a treatment onto a damaged heart? They really did spray a solution containing the active agent directly onto the heart. And they note that the sprayer design anticipated "future minimally invasive surgical procedures." The authors compare their treatment mode to using patches; spraying gives a coating that conforms to the heart shape.
How does the treatment work? That's not at all clear. Survival of heart tissue following a heart attack is rather mysterious, and certainly generally not good (in mammals). It is plausible that the gold is important here, perhaps in providing a more conductive surface, and that a key role of the protein is to organize the gold nanoparticles. It is also possible that there are multiple effects, and that the overall effect seen above is due to synergy. The data might actually hint at this.
Whatever is going on, there is evidence it works, in the mouse model. The material itself is practical to make, and the delivery method is thought to be practical, if not ideal. Thus the work here deserves follow-up.
"In summary, we have demonstrated that by combining peptide design and an on-the-spot application, a therapeutic approach was developed that allows nanomolar concentrations of nanogold to restore cardiac function in a clinically relevant animal model of myocardial infarction. However, the mechanism of action by which the application of nanogold restores cardiac function postmyocardial infarction remains to be fully elucidated, including delivery of the therapy at different time points after heart injury. Our findings illustrate how peptide engineering can finely tune the properties of nanomaterials for use in therapeutic applications that, in our case, improved cardiac contractility and electrical signal propagation." That is the first paragraph of the Conclusions section of the article.
News stories:
* Spray of gold nanoparticles can potentially treat heart disease. (Nanowerk News (University of Ottawa), March 15, 2022.)
* The golden future of cardiac tissue repair. (The Beat, University of Ottawa Heart Institute, March 2022.) Interesting name for a newsletter from a heart institute.
The article, which is open access: Nanoengineered Sprayable Therapy for Treating Myocardial Infarction. (Marcelo Muñoz et al, ACS Nano 16:3522, March 22, 2022.)
A post about work involving the use of artificial heart attacks in lab rodents (rats, in this case); it is a standard lab system. Treating a heart attack using a microneedle patch (January 11, 2019).
There is more about regeneration on my page Biotechnology in the News (BITN) for Cloning and stem cells. It includes an extensive list of related Musings posts, including some on heart issues.
Previous post about gold nanoparticles: Super-resolution microscopy -- without special labels (May 15, 2022). Just two days ago. Any connection? One is on analysis, one on use.
More gold... Prospecting for gold -- with help from the little ones (March 1, 2013). Includes a list of gold-related posts.
Briefly noted... How long can you track magpies fitted with a GPS device?
May 16, 2022
Until they remove them. That may take only a few minutes, because they help each other remove the devices. At one level, it is just a fun story, but there may be interesting implications for our understanding of these smart birds.
* News stories. Both link to the article, which is open access.
- Australian birds opt out of experiments. By removing tracking devices? -- "The birds outsmarted us". (Ameya Paleja, Interesting Engineering, February 23, 2022.) Also links to the following...
- Altruism in birds? Magpies have outwitted scientists by helping each other remove tracking devices. (Dominique Potvin, The Conversation, February 21, 2022.) By one of the authors of the article.
* More about the corvids: Ravens: planning for the future? (September 11, 2017). Links to more.
Super-resolution microscopy -- without special labels
May 15, 2022
A new article reports another advance in high-resolution microscopy. In this case, the key development is that the procedure does not require any special pre-treatment of the sample.
The following figure shows examples of what could be seen...

|
Each part shows two images of the same material, which consists of tiny gold nanoparticles -- sometimes, groups of them.
The top images are from light microscopy (LM), using the method developed here. They are labeled 'laser scan images'. The bottom images are from (scanning) electron microscopy ((S)EM).
The LM images are not as good as the EM images. But they are very good. The EM images clearly show the particles. The particles are not so clear in the LM images, but they can be resolved by mathematical analysis of the image. The red circles in each EM image show the particles as deduced from the LM image. Not bad.
|
The scale bars are 532 nm -- the wavelength of the light they used.
The authors note that the LM images were taken first, because the EM imaging damaged the samples.
This is part of Figure 3 from the article.
|
The authors claim resolution of "several nanometers".
The resolution of ordinary image formation is limited to about half the wavelength of the light used. In this case, that would be about 266 nm.
There have been many developments in high-resolution microscopy over recent years. The big idea is to avoid that ordinary image formation, which loses information. Instead, the light is analyzed in more detail. Often, it is necessary to first modify the material, for example, to make it fluorescent.
In this case, the system starts with laser scanning -- high intensity monochromatic light, localized. Then there is complex analysis of the light, including polarization and scattering. The details are worked out to be useful; what matters, for the moment, is that the microscope is based on complex analysis of the light, not simply ordinary image formation. Importantly in this work, no pre-treatment of the sample is used.
The authors note limitations of what they have done here, and will try to extend the usefulness of the approach.
News stories:
* Researchers demonstrate label-free super-resolution microscopy. (Nanowerk News (from the journal), April 21, 2022.)
* Noninvasive Imaging Method Measures Below the Diffraction Limit. (Photonics Media, April 29, 2022.)
The article, which is open access: Sub-diffraction-limit Fourier-plane laser scanning microscopy. (Jörg S Eismann & Peter Banzer, Optica 9:455, May 2022.)
More about advances in microscopy:
* Visualizing atoms with electron ptychography -- approaching the theoretical resolution (June 5, 2021).
* Expansion microscopy: making an object bigger can make it easier to see (February 23, 2015). Includes a link to the recent Nobel Prize for super-res microscopy.
There is a section of my page Internet resources: Biology - Miscellaneous on Microscopy. It includes a list of related Musings posts.
Next post about gold nanoparticles: A gold-based spray to treat heart damage (May 17, 2022). Two days later; just above.
May 11, 2022
Briefly noted... Does the word 'rough' sound r-r-r-rough?
May 11, 2022
It's that trill. And it's not just an accident of English. A recent article analyzed more than 300 languages, from many language families around the world. Not all languages have the trilled-r sound, but for languages that do, it is far more likely to be in the word for rough than in random adjectives or specifically the word for smooth. It is a connection between sound and touch. For the Indo-European language family, the use of trilled-r in 'rough'-like words dates back at least 6,000 years.
* News story: 'Rough' Words Feature a Trill Sound in Languages Around the Globe. (Neuroscience News (University of Birmingham), January 21, 2022.) The main figure summarizes some of the findings: there are far more red points than blue. Links to the article, which is open access.
* Also see: Mountains and human language? (June 28, 2013). Links to more about language.
Briefly noted... Dinosaur extinction: What time of year was it?
May 10, 2022
It's spring (in the North). An appropriate time, according to a recent article, to discuss dinosaur extinction. A team of scientists examined fossil fish found at the geological boundary marking the extinction of the (non-avian) dinosaurs. The fish were likely killed within a hour or so of the event, and they are remarkably well preserved. Detailed examination of the fish indicates that they died during the spring; yes, some fish have annual growth rings, too. That's boreal (northern hemisphere) spring; it was autumn in the South. Simply getting that timing leads to ideas about why some organisms may have been more vulnerable. Although the logic of the work is straightforward, the details of the analysis are not easy to follow. The news stories should give you the ideas and some context.
* News stories. The last two both link to the article, which is open access.
- The Last Day of the Mesozoic - It was Boreal Spring. (Everything Dinosaur, February 24, 2022.) Notes a study from last year which came to a similar conclusion, from different evidence.
- The reign of the dinosaurs ended in spring. (Geology Page (Uppsala University), February 27, 2022.)
- The Reign of the Dinosaurs Ended in Spring: Revelations From Bones of Fish That Died When the Asteroid Hit. (SciTechDaily (European Synchrotron Radiation Facility), February 23, 2022.)
* Also see...
- Modeling the tsunami from the asteroid impact that caused the dinosaur extinction (October 16, 2022).
- What caused the extinction of the dinosaurs: Another new twist? (January 26, 2016).
- Do animal bones have something like annual growth rings? (August 7, 2012).
Environmental sensors that can be dispersed by the wind and land upright
May 9, 2022
Engineers are interested in developing ways to monitor the environment. Spreading tiny sensor devices around would be good.
As one approach, engineers turn to nature for inspiration. Various plant seeds are dispersed by the wind. Engineers learn how this works, and design artificial devices to use similar approaches. Musings recently noted an example of tiny flying devices inspired by maple seeds [link at the end].
A recent article reports another example, inspired by dandelion seeds. Dandelion seeds drift in the wind while falling, and land upright. That behavior served as the inspiration for designing a tiny environmental sensor, which can be distributed around by the wind. The ability to land upright means it can work with solar energy, rather than heavier batteries.
Look...

|
The figure shows a dandelion seed and an example of the devices developed in the current work.
Both have a canopy, which serves to keep the thing airborne, and a weight at the bottom, which helps keep it right-side-up.
This is Figure 1a from the article.
|
Nature worked out the dandelion seed. The scientists here had to explore the details to make an effective wind-dispersed sensor device. They explored a range of canopy sizes and structures. Of course, their device must be able to carry useful equipment, some of which is on the canopy and some of which is at the bottom.
Note that the canopy of the sensor device has a ring around it. That is an example of how the scientists built on nature's seed to make the current device. The ring adds stability to the device, which is about 30 times heavier than the dandelion seed.
One important design goal was that the device land upright, so the solar cell would work efficiently. The following figure shows how well this worked out...
The figure shows the percent of the devices that landed upright (y-axis), vs the size of the canopy structure (x-axis), and the "fill %" (color coding; see key at the right).
The results suggest a good size range for devices that reliably land upright. (It's hard to tell much about the fill % from this graph alone.)
This is Figure 2f from the article.
|

|
The device is good over about a 50-100 meter range. That is the range over which light-moderate wind will disperse it, and the range its wireless signals can reach.
News stories:
* Tiny battery-free devices float in the wind like dandelion seeds. (Sarah McQuate, University of Washington, March 16, 2022.)
* 'Floating Sensors' Spread Like Dandelion Seeds -- Dropped by a drone, the battery-free devices hover 100 meters in the air. (Tech Briefs, April 14, 2022.) Includes an interview with the first author, Vikram Iyer.
The article: Wind dispersal of battery-free wireless devices. (Vikram Iyer et al, Nature 603:427, March 17, 2022.)
Background post: The smallest manmade flying devices (December 12, 2021). The article of this earlier post is reference 23 of the current article.
Also see: Smart dust: A central nervous system for the earth (July 20, 2010).
For more about bio-inspiration, see my Biotechnology in the News (BITN) topic Bio-inspiration (biomimetics). It includes a listing of Musings posts in the area.
Pupil dilation as an indicator of visual imagination
May 7, 2022
The pupil of the eye gets larger (dilated) when there is less light. That is familiar.
Here are some data from a recent article -- with a twist...
The y-axis shows the change in pupil diameter, under various conditions. Blue data is for dark conditions, red is for bright conditions.
In the first segment (1-6 seconds), called perception, one or another condition was provided. The results show that the pupils got larger under dark conditions, smaller under bright conditions. As expected.
In the next segment, called rest, the stimulus was removed. Over a few seconds, the pupils of those from both types of stimuli became similar.
In the third segment, called imagery, each person was asked to imagine what they had perceived in the first segment. Those who had perceived and now imagined a dark stimulus got larger pupils than those who had perceived and now imagined a bright stimulus.
This is Figure 1B from the article.
|
That is, light intensity can cause changes in pupil size not only during direct observation but during mental imagery. In the latter case, the pupil change is a physical manifestation of an act of imagination, and one that can be objectively measured.
That is the first major finding of the work.
Here are the results of a similar experiment with another group of people...
The results are generally similar to the top set, with one important exception: there is no difference between the dark ad bright groups during the imagery segment.
The people in this test have aphantasia, as labeled in the title of the figure. That means they lack visual imagination.
This is Figure 3A from the article.
|
People self-report having aphantasia; there has been no test for it. The current work would seem to provide such a test, and to validate the self-reported condition.
Overall, the work here shows that a simple physical measurement may yield information about how the imagination works.
News stories:
* Windows to the soul: Pupils reveal 'aphantasia' -- the absence of visual imagination. (Science Daily (University of New South Wales), April 20, 2022.)
* Pupil Size May Reflect the Strength of Your Imagination -- Pupil size indicates the vividness of imagination and mental imagery strength. (Christopher Bergland, Psychology Today, May 1, 2022.) Includes some discussion of another article in the field.
The article, which is open access: The pupillary light response as a physiological index of aphantasia, sensory and phenomenological imagery strength. (Lachlan Kay et al, eLife 11:e72484, March 31, 2022.)
Another post about pupil dilation: Bright lights and pupil contraction (March 2, 2012). It, too, suggests that dilation is more complex then we might have thought.
May 5, 2022
Briefly noted... Progress toward making a more people-friendly (hypoallergenic) cat
May 5, 2022
Many people are allergic to cats. The major allergen, called fel d, is known. The question is, can we do anything about it? A recent article reports some explorations, including the use of CRISPR to inactivate one of the genes for fel d. Examination of the gene sequences for a wide range of cats, including several of the "big cats", shows that the gene is highly variable, perhaps suggesting it is not essential. The variability of expression in domestic cats is in agreement. That might suggest that eliminating the protein would be of no consequence to the cat. The work here is with cell cultures, and is successful. It is not clear where this leads. The authors do not think it is worthwhile to breed allergen-free cats. Instead, they envision a treatment to reduce the allergen levels of adult cats. We'll see. In any case, it is a fun article, and the issues are worth thinking about.
* News story: Scientists Are Inching Closer to Creating Truly Hypoallergenic Cats -- The gene-editing technology CRISPR could knock out a protein that's the major source of cat allergies, a new study says. (Ed Cara, Gizmodo, March 28, 2022.) Links to the article, which is open access.
* Here is an earlier news story, from last Fall. It gives an overview of various approaches to reducing the amount of cat allergen. It includes the current approach, but this story predates the current article. The Next Weird Way We're Changing Cats -- What if you could make your cat hypoallergenic with biotechnology? (Sarah Zhang, The Atlantic, November 5, 2021.)
* The article, which is open access: Evolutionary Biology and Gene Editing of Cat Allergen, Fel d 1. (Nicole F Brackett et al, CRISPR Journal 5:213, April 2022.)
* Posts about CRISPR are listed at: CRISPR: an overview (February 15, 2015).
* Previous mention of cat allergies: Pet Diary (September 25, 2009).
Atomic clock measurements of the difference in gravity over one millimeter
May 4, 2022
Gravity -- the gravitational field of a massive object -- distorts space-time. As a result, clocks run more slowly at higher gravity; clocks run more slowly on the surface of Earth than up above.
Look...
Part a (left) shows an experiment done 50 years ago. Clocks on the ground and in a high-altitude spacecraft were compared. It was found that the clock on the ground ran more slowly; the Earth clock reports less time elapsed, as predicted.
Parts b and c show two experiments reported in a pair of recent articles. Both involved the use of clouds of strontium atoms as atomic clocks. In one case, two clouds a centimeter apart were compared. In the other case, the top and bottom of a single cloud, a millimeter high, were compared.
In both cases, the predicted effect was seen: the lower clock ran more slowly.
This is Figure 1 from the news story accompanying the article (Khabarova).
|
What's remarkable here is the sensitivity of the experimental systems -- the quality of the clocks.
Such atomic clocks operate by measuring the frequency of a particular transition within the atom. The observed time measurement may be affected by the gravitational field.
Here is an example of the results, from article 1...

Part a (left) shows results from a single test. The y-axis shows the measured frequency -- as the difference from the mean value. Note that 1 on the y-axis scale means 1 part in 1019. Green is for the raw data; purple is for corrected data.
The x-axis is for the elevation of the measurement; it covers about a one millimeter range.
The graph includes a best-fit line through the data. You can see that the line has a slight negative slope. The frequency varies with elevation.
Part b summarizes the results from 14 tests. Each point summarizes one test, with a mean and error bar. The black line at y = -1 shows the mean of those 14 tests, along with error bars. The red line is the expected value, from theory.
The left-hand y-axis is much like that in part a, but is now the change in frequency per millimeter of elevation. The right-hand y-axis is for the same results, but now reported as a change in gravity.
This is Figure 3 from article 1 (Bothwell et al).
|
Overall, the results show that there is a change in the observed frequency of the clock by about 1 part in 1019 per millimeter of elevation. The measured result is very close to what is predicted.
For perspective... The difference in gravity between the head and feet of a child (height 1 meter) is a thousand times larger than what is observed here.
Is this for real? The point of the work is to push the limits. That is what experimental physicists do. The clocks here are about 50 times better than the previous best.
As for the results... Well, they were as expected. But an interesting point is developing the ability to measure gravity over extremely short distances. Is this a step toward unifying the theory of gravity, best understood at the large scale, with quantum mechanics, best understood at the small scale?
News stories:
* New Atomic Clocks Measure Time Dilation of Einstein's General Relativity at Millimeter Scale. (SciTechDaily (National Institute of Standards and Technology (NIST)), February 16, 2022..) Article 1.
* Ultraprecise Atomic Clock Poised for New Physics Discoveries - Loses Just One Second Every 300 Billion Years. (SciTechDaily (University of Wisconsin-Madison), February 20, 2022.) Article 2. Briefly mentions article 1.
* News story accompanying the articles: Metrology: Atomic clouds stabilized to measure dilation of time -- Tests of relativity once required accurate clocks separated by thousands of kilometres. Optical techniques have now made such tests possible in an atomic cluster measuring no more than one millimetre in size. (Ksenia Khabarova, Nature 602:391, February 17, 2022.)
* Two articles:
1) Resolving the gravitational redshift across a millimetre-scale atomic sample. (Tobias Bothwell et al, Nature 602:420, February 17, 2022.)
2) Differential clock comparisons with a multiplexed optical lattice clock. (Xin Zheng et al, Nature 602:425, February 17, 2022.)
More about gravity and time: Which is older, the center of the Earth or the surface? (September 7, 2016). Links to more, broadly about gravity.
More about time: A nuclear clock (October 16, 2024).
More strontium...
* Atoms within atoms? (May 25, 2018).
* Revealing the alabaster sources of ancient artists (March 7, 2018).
Design criteria for mRNA: the start codon
May 2, 2022
The start codon -- the first codon of a messenger RNA that is translated -- is AUG. It codes for methionine.
Of course, the real world is more complex than that. Other codons also serve as start codons, at lower frequency. The start codons vary in the first base; that is, they are all of the form NUG, where N stands for any base. Whichever start codon is used, it codes for methionine. The details vary, especially for prokaryotes vs eukaryotes, which use rather different procedures for initiation of protein synthesis.
A new article explores the effect of modified bases in the mRNA on the use of various start codons. The findings offer some clues for the details of how initiation works, but also has implications for how mRNA made for therapeutic use might be made better.
The following figure summarizes some of the findings...
The general plan is that various artificial genes were made, and tested to see how well they worked -- in human cells in lab culture. Each construct led to production of green fluorescent protein (GFP); the amount of GFP made is easily measured, and was used to evaluate the level of initiation.
Eleven sequences were tested. They are listed along the left side, and numbered at the right, along with the sequence of the initiation codon and nearby region.
For each sequence, there were two variations. As a control, the RNA was made using the natural (standard) bases. And then the main experimental variable... the mRNA was also made using two modified bases: 5-methylcytosine (5mC) and pseudouridine (PsU or ψ). These changes do not change the general nature of the base pairing, but they may affect details of interactions.
Let's look at some of the results...
Set 1 is labeled Kozak AUG. That refers to the consensus sequence for a good start codon (as originally elucidated by Marilyn Kozak). The result for natural bases is set to 100%. The result with modified bases is the same.
Set 2 is labeled Kozak GUG. Same as #1, except that the AUG start codon has been changed to GUG. In the sequences shown at the right, the start codon is underlined, and the change is shown in red. With this start sequence, it now functions poorly (about 10%) with natural bases, but rather well (about 60%) with the modified bases.
Set 3 introduces the actual initiation region from a specific human gene, called NAT1. This gene has a GUG start codon. The results show that it responds similarly to the Kozak GUG case (#2).
In set 5, that GUG start codon has been changed to AUG. It now behaves like the Kozak AUG case (#1).
Skip down to set 10. That NAT1 start codon is now UUG. It functions much like the GUG cases, working well only with the modified bases.
NAT stands for N-acetyltransferase.
This is Figure 1C from the article.
|
The general picture is that the modified bases in the mRNA make little difference when there is a standard AUG start codon. However, genes with other start codons, which are normally translated poorly, are translated much better with the modified bases.
mRNA is being explored as a type of drug. The COVID vaccines are an example. Pseudouridine (actually, 1-methylpseudouridine) is used in COVID mRNA vaccines, because it reduced the immune response to the RNA. The new work opens up further possibilities for how modified bases can be a tool in designing therapeutic mRNAs. For example, an mRNA designed with a UUG start codon and made with the modified bases of the current work will be translated rather efficiently. However, if it gets copied, it won't be translated much, because UUG is a very poor start codon on its own.
Notes...
Both modified bases used here are found naturally, including in mRNA.
The authors provide some information on how the modified bases affect initiation, by altering the interaction with one of the initiation factors. This is based on calculations using computer models of the initiation mechanism.
News stories:
* Chemical markers that may unlock future uses of messenger RNA. (Ria Kakkad, Drug Target Review, April 28, 2022.)
* Scientists identify chemical markers that may unlock future therapeutic uses of mRNA. (Nanowerk News (Hiroshima University), April 26, 2022.)
The article, which is open access: Translational recoding by chemical modification of non-AUG start codon ribonucleotide bases. (Yoshihiko Fujita et al, Science Advances 8:eabm8501, April 8, 2022.)
Another post exploring how protein synthesis can be tweaked... Ribosomes with subunits that are tethered together (October 5, 2015).
For some background on GFP: Nobel prizes (October 8, 2008). Links to more.
Top of page
The main page for current items is Musings.
The first archive page is Musings Archive.
E-mail announcements -- information and sign-up: e-mail announcements. The list is being used only occasionally.
Contact information
Site home page
Last update: May 13, 2026