Bottom of page; return links and contact information
| 1. |

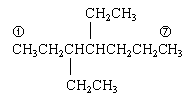
| Strategy: Draw heptane, the "root" or "parent" compound as the name indicates. Number the C atoms, 1 to 7. (The circled numbers show the chain numbering.) Then attach the indicated groups. Finally, add enough H so that each C has four bonds. |
2.
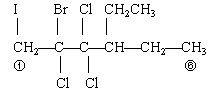
3. No. Isomers have the same molecular formula, but different structures. These two compounds do not have the same molecular formula.
Be sure you have the correct chemicals before trying to answer the question. Don't be misled by the names.
| 4. |
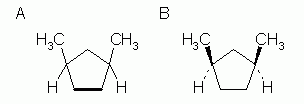
| Both methyl groups are shown on the same side (up, but down would be fine); the given compound is "cis". |
| ||
5. Hexane. The two chemicals are of the same type (i.e., both are alkanes), and hexane is bigger = heavier. Therefore, there are greater London forces between the molecules.
6. n-butane has the higher boiling point (BP). The two chemicals are of the same type (alkanes) and they have the same mass. So what gives them different BP? Their shapes. The more tightly two molecules can stick together, the harder it is to break them apart, thus the higher the BP. Linear molecules can stick together better than branched ones, which tend to look like balls.
I suggest you review Ouellette Fig 2.3 and accompanying text.
Simply quoting the boiling points of these compounds misses the purpose of the question, and would receive no credit on a test. The question discusses an issue, and asks for a reason. Of course, if you do happen to know which has the higher BP, that can help guide you, but per se makes no intellectual contribution.
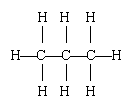
The two compounds stated are the only two compounds with the molecular formula C4H10.
7. a.
b. There are only two: 1-chloropropane (with a Cl on a terminal C), and 2-chloropropane (with a Cl on the middle C).
If you think you have more than 2... I suggest that you make a model of each. See if you can rotate one model so that it looks identical to another. If so, the two structures you thought were different are actually the same.
c. 6. Replacing any one of the H on the -CH3 group at either end gives the same result.
d & e.
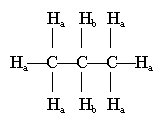
8. The chloropropane is somewhat more polar than propane, so should be somewhat more soluble. (In this case, neither is very soluble. But it is true that the chloro compound is more soluble.)
9. 2,2-dimethylpropane (common name: neopentane).
A quaternary C has 4 other C attached to it. Ouellette, p 71. It is also on my page Terms: Primary, Secondary, Tertiary, Quaternary.
| 10. |
| It is C4I2. No hydrogens. |
So how did we get that? Why are there no numbers in the name to tell us where the triple bonds or I atoms are? The name is diiodobutadiyne. Let's break that down: di iodo buta di yne. Four C; two triple bonds; two I. So lay out 4 C atoms: C C C C. Now, two triple bonds. Where? Triple bonds cannot be next to each other; that is, one C cannot have two triple bonds. Therefore the only possible place for the triple bonds is at the ends (i.e., 1,3-butadiyne). Now, look for places to put I -- or H. Each C forms 4 bonds; the only places left are at the ends, so put the two I atoms there. That's it. There are no more bonds available on the C atoms, so there are no H.
This compound is an example where numbers ("locants") are not needed to specify the location of groups, because there is only one possibility. If you were asked to name this compound, you might well include numbers, without noticing that they are not needed in this special case. That's ok. But here you are given the name, and should be able to figure out where things go. For more about the special cases where numbers are not needed, see my page on Omitting numbers. Remember, if in doubt, include numbers.
The quiz Quiz list Organic/Biochem (X402) home page
Contact information Site home page
Last update: August 22, 2019