Ouellette 2/e Ch 4.
Answer key
Bottom of page; return links and contact information
Closed book.
1. Draw the structures of all possible (mono)chloro-substituted forms of 1-butene. Name each isomer (IUPAC preferred).
2. Draw the structure of an isomer of the above compounds which is not an alkene. Name the compound.
3. How could you "quickly and easily" distinguish the compound you drew in #2 from its isomers in #1? (Note: The Cl in these compounds is not relevant, at least qualitatively.)
4. Show how you could make 2-bromo-2-methylbutane starting from an alkene. (You choose the alkene, and you can use "standard reagents".)
5. Give an example of a compound in which one particular C atom has more than one multiple bond (double or triple bond). Name the compound.
6. Write a balanced chemical equation for the complete combustion of a cycloalkene.
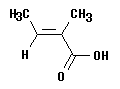
7. Consider the following alkene (actually, an alkenoic acid):

a. In what sense could you say that the term "cis" applies to this structure?
b. In what sense could you say that the term "trans" applies to this structure? (You need to know one fact that hasn't officially come up yet... Carbon #1 is the C at lower right, with the oxygens.)
c. What is the best way to designate which isomer this is in an IUPAC name? (Confused? You shouldn't be. This is E-Z!)
d. (optional, because we have not covered naming of acids; the acid is designated by the suffix "-oic acid") What is the IUPAC name?
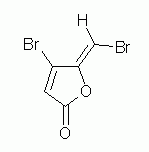
8. Assign E or Z to each double bond in the following compound. Explain.
9. Fill in each blank with an appropriate number. sp2 orbitals are formed by hybridizing [how many] ___ s orbital(s) and ___ p orbital(s). Since ___ orbitals (total) are used in making the hybrid orbitals, the number of sp2 orbitals obtained is ___. Therefore, sp2 orbitals are appropriate when there are ___ "things" (atoms or lone pairs) attached to a central atom. The bond angle that is typical of sp2 hybridization is ____ degrees.
Answer key Quiz list Organic/Biochem (X402) home page
Contact information Site home page
Last update: August 29, 2019