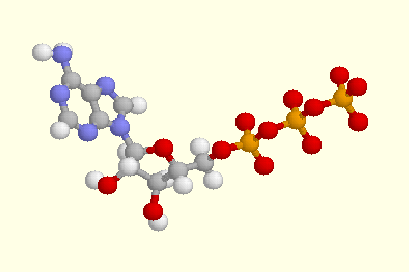

The model above shows a molecule of adenosine triphosphate, commonly known as ATP.
Bottom of page; return links and contact information
ATP is a common "energy currency" in all living organisms. The organism burns food, and stores some of the energy from the "burning" in the bonds of the ATP molecule. Reactions of the ATP that lead to bonds storing less energy are used to supply energy to other reactions in the organism.
In the model above, the grey balls represent carbon (C) atoms, white balls represent hydrogen (H) atoms, blue balls represent nitrogen (N) atoms, red balls represent oxygen (O) atoms, and orange balls represent phosphorus (P) atoms.
One can think about the ATP molecule as having three main parts:
* The left (two fused rings with lots of blue balls, for N) is adenine. Adenine is a base, in the proper chemical sense; adenine is also one of the common bases ("A") in ribonucleic acid (RNA) and deoxyribonucleic acid (DNA).
* The middle (ring with three red balls, for O) is ribose, a sugar. Ribose is the sugar of RNA, as well as of ATP. (The sugar in DNA is deoxyribose; this is just like ribose, except one O is absent.)
* The right side of the model (orange and red balls, for P and O) shows the three phosphate groups. One is connected to the ribose sugar by an ester linkage. The remaining two phosphates are attached to the preceding phosphate by an acid anhydride linkage. The two acid anhydride linkages are the key places where energy is "stored" in ATP.
All of the issues mentioned here are discussed in an introductory course in organic and biochemistry.
Organic/Biochem (X402) home page
Contact information Site home page
Last update: December 9, 2023